��Ŀ����
��1��N2��g����3H2��g��  2NH3��g������H����94.4 kJ��mol��1������ʱ����ϵ�и�����Ũ����ʱ��仯��������ͼ��ʾ��
2NH3��g������H����94.4 kJ��mol��1������ʱ����ϵ�и�����Ũ����ʱ��仯��������ͼ��ʾ��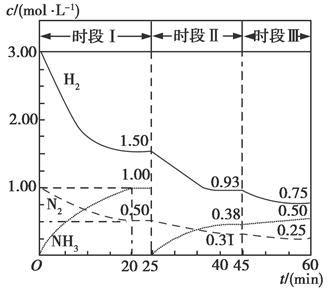
����1 L�����з�����Ӧ��ǰ20 min�ڣ�v��NH3����________���ų�������Ϊ________��
��25 minʱ��ȡ�Ĵ�ʩ��_______________________��
��ʱ�������·�Ӧ��ƽ�ⳣ������ʽΪ________���þ������ݱ�ʾ����
��2���糧�������뵪������Ӧ�٣�4NH3��g����6NO��g�� 5N2��g����6H2O��g��������Ӧ�ڣ�2NH3��g����8NO ��g��
5N2��g����6H2O��g��������Ӧ�ڣ�2NH3��g����8NO ��g��  5N2O��g����3H2O��g������H��0�����ƽ��������N2��N2O�������¶ȵĹ�ϵ��ͼ����400��600 Kʱ��ƽ��������N2�������¶ȵı仯������________���������ֱ仯���ɵ�ԭ����________���δ������һ��ԭ��
5N2O��g����3H2O��g������H��0�����ƽ��������N2��N2O�������¶ȵĹ�ϵ��ͼ����400��600 Kʱ��ƽ��������N2�������¶ȵı仯������________���������ֱ仯���ɵ�ԭ����________���δ������һ��ԭ��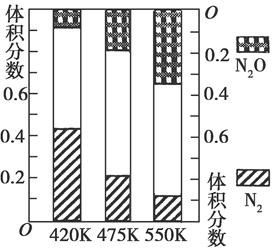
��3��ֱ�ӹ���ʽȼ�ϵ������NaOH��ҺΪ����ʵġ���ط�ӦΪ4NH3��3O2=2N2��6H2O�����缫��ӦʽΪ________��
��1����0.050 mol����L��min����1��47.2 kJ
�ڽ�NH3�ӷ�Ӧ��ϵ�з����ȥ
��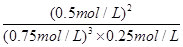
��2�����¶����ߣ�N2�ĺ������͡�����ӦΪ���ȷ�Ӧ������ʹ����Ӧ��ƽ�����ƣ����߸���ӦΪ���ȷ�Ӧ������ʹ����Ӧ��ƽ�����ƣ�������NH3��NOŨ�ȣ�ʹ����Ӧ��ƽ�����ƣ�
��3��2NH3��6OH����6e��=N2��6H2O
����

 �̲�ȫ���ִʾ�ƪϵ�д�
�̲�ȫ���ִʾ�ƪϵ�д���ӦA��3B=2C��2D�����ֲ�ͬ����µķ�Ӧ���ʿɷֱ�Ϊ
��v
| A����0.15 mol/(L��s)����v | B����0.6 mol/(L��s)����v | C����0.4 mol/(L��s)����v | D����0.45 mol/(L��s) |
��ijһ�ݻ�Ϊ5 L���ܱ������ڣ�����0.2mol ��CO��0.2mol��H2O��g�����ڴ������ڵ������¸��¼��ȣ��������·�Ӧ��CO��g����H2O��g�� CO2��g����H2��g������Ӧ�ų���������Ӧ��CO2��Ũ����ʱ��仯���������ͼ��ʾ��
CO2��g����H2��g������Ӧ�ų���������Ӧ��CO2��Ũ����ʱ��仯���������ͼ��ʾ��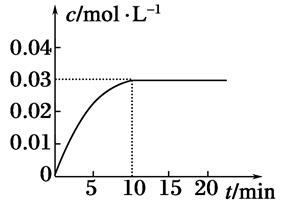
��1������ͼ�����ݣ��ӷ�Ӧ��ʼ���ﵽƽ��ʱ��CO�Ļ�ѧ��Ӧ����Ϊ________����Ӧƽ��ʱc��H2����________��
��2���жϸ÷�Ӧ�ﵽƽ���������________������ţ���
| A��CO���ٵĻ�ѧ��Ӧ���ʺ�CO2���ٵĻ�ѧ��Ӧ������� |
| B��CO��H2O��CO2��H2��Ũ�ȶ���� |
| C��CO��H2O��CO2��H2��Ũ�ȶ����ٷ����仯 |
| D�������淴Ӧ���ʶ�Ϊ�� |
����þ���仯���ﲻ���ڹ���������ҪӦ�ã��ڻ�ѧ�о���Ҳ�й㷺Ӧ�á�
��1��ijͬѧ�о���Ӧ����ʱ�����·�����ȡһ��þ������ɰֽ��ȥ���������Ĥ��ʹ����þ����һ�������ᷴӦ����H2�����뷴Ӧʱ��Ĺ�ϵ������ͼ��ʾ��þ�����ᷴӦ�����ӷ���ʽΪ ����ǰ4min�ڣ�þ��������ķ�Ӧ�����ӿ죬��4 min֮��Ӧ�����������������ԭ��_____________��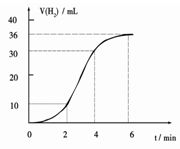
��2����������Mg(OH)2����Һ�м��������ı����Ȼ����Һ��������ȫ�ܽ⣬д��NH4Cl������ҺʹMg(OH)2����Һ�ܽ�����ӷ���ʽ ��
��3��Mg��Al���γ�ԭ���:
| ��� | �缫���� | �������Һ | ������������ |
| 1 | Mg��Al | ϡ���� | Mg����Al |
| 2 | Mg��Al | NaOH��Һ | |
�����ϱ��е�ʵ�����������������:
��ʵ��1��MgΪ ����
��ʵ��2�е����������� ��AlΪ ����Ϊʲô?����������������
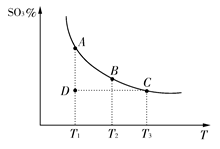
 2SO3(g)�������ϵ��SO3�İٷֺ������¶ȵĹ�ϵ����ͼ��ʾ(�������κ�һ�㶼��ʾƽ��״̬)������ͼʾ�ش��������⣺
2SO3(g)�������ϵ��SO3�İٷֺ������¶ȵĹ�ϵ����ͼ��ʾ(�������κ�һ�㶼��ʾƽ��״̬)������ͼʾ�ش��������⣺
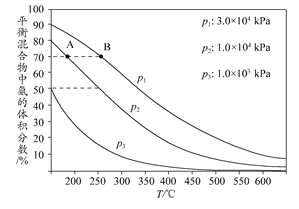

 2NH3 ��H<0���������¶Ȳ��䣬ij��ȤС��ͬѧ��÷�Ӧ������������ѹǿ��ʱ��仯��ͼ��ʾ��8 min�ڷ���NH3��ƽ����������Ϊ mol��L-1��min-1��
2NH3 ��H<0���������¶Ȳ��䣬ij��ȤС��ͬѧ��÷�Ӧ������������ѹǿ��ʱ��仯��ͼ��ʾ��8 min�ڷ���NH3��ƽ����������Ϊ mol��L-1��min-1��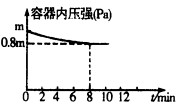

 H=-akJ��mol-1
H=-akJ��mol-1 2Fe2++I2(ˮ��Һ)��
2Fe2++I2(ˮ��Һ)��