��Ŀ����
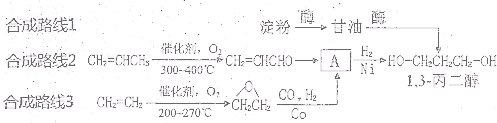
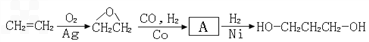
1��3-���������������߷��Ӳ���PTT����Ҫԭ�ϣ�Ŀǰ������·�������¼��֣�
��֪�������������CH2��COOC2H5��2���ܷ������·�Ӧ��
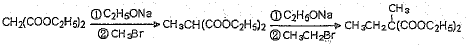
��1���л���A���еĹ�����Ϊ ��
��2���Ӻϳ�ԭ����Դ�ĽǶȿ�������Ϊ����з�չǰ����·���� ����1��2��3���������� ��
��3����1��3-��������Ա������ᣨ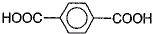 ��Ϊԭ�Ͽ��Ժϳɾ���PTT��д���仯ѧ����ʽ ��
��Ϊԭ�Ͽ��Ժϳɾ���PTT��д���仯ѧ����ʽ ��
�Ա������������1��3-���������Ҵ�Ϊԭ�ϣ�������ѡ���ϳ�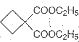 ����ת��Ϊ��
����ת��Ϊ��
��4��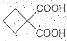 ��ͬ���칹�岻�������� ��
��ͬ���칹�岻�������� ��
a���� b���� c��ȩ d����
��5��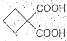 Ҫ�ϳɣ������Ⱥϳ���Щ���ʣ����úϳɸ����ʵĻ�ѧ����ʽ�ش� ��
Ҫ�ϳɣ������Ⱥϳ���Щ���ʣ����úϳɸ����ʵĻ�ѧ����ʽ�ش� ��

��֪�������������CH2��COOC2H5��2���ܷ������·�Ӧ��
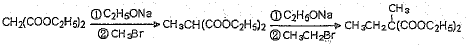
��1���л���A���еĹ�����Ϊ
��2���Ӻϳ�ԭ����Դ�ĽǶȿ�������Ϊ����з�չǰ����·����
��3����1��3-��������Ա������ᣨ
 ��Ϊԭ�Ͽ��Ժϳɾ���PTT��д���仯ѧ����ʽ
��Ϊԭ�Ͽ��Ժϳɾ���PTT��д���仯ѧ����ʽ�Ա������������1��3-���������Ҵ�Ϊԭ�ϣ�������ѡ���ϳ�
 ����ת��Ϊ��
����ת��Ϊ����4��
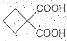 ��ͬ���칹�岻��������
��ͬ���칹�岻��������a���� b���� c��ȩ d����
��5��
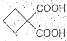 Ҫ�ϳɣ������Ⱥϳ���Щ���ʣ����úϳɸ����ʵĻ�ѧ����ʽ�ش�
Ҫ�ϳɣ������Ⱥϳ���Щ���ʣ����úϳɸ����ʵĻ�ѧ����ʽ�ش���������1��A�����������ӳɷ�Ӧ����1��3-��������A�к��в����ͼ�������������CO�����������ӳɷ�Ӧ����A��A�к���-CHO����ϩȩת��ΪA��Ӧ����ˮ�����ӳɷ�Ӧ����AΪHOCH2CH2CHO��
��2������ԭ�ϵ��Ƿ�Ϊ��������Դ�����жϣ�
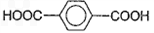
��3��1��3-��������Ա�������ɷ������۷�Ӧ���Դ˿���д��Ӧ�Ļ�ѧ����ʽ��
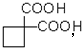
��4��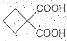 ���Ͷ�Ϊ3�����ຬ�б������䲻���Ͷ�����Ϊ4������ͬ���칹�岻����Ϊ���ࣻ
���Ͷ�Ϊ3�����ຬ�б������䲻���Ͷ�����Ϊ4������ͬ���칹�岻����Ϊ���ࣻ
��5���������⣬Ӧ������C2H5ONa��CH2BrCH2CH2Br��Ȼ����������������Ӧ������Ŀ���
��2������ԭ�ϵ��Ƿ�Ϊ��������Դ�����жϣ�
��3��1��3-��������Ա�������ɷ������۷�Ӧ���Դ˿���д��Ӧ�Ļ�ѧ����ʽ��
��4��
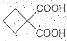 ���Ͷ�Ϊ3�����ຬ�б������䲻���Ͷ�����Ϊ4������ͬ���칹�岻����Ϊ���ࣻ
���Ͷ�Ϊ3�����ຬ�б������䲻���Ͷ�����Ϊ4������ͬ���칹�岻����Ϊ���ࣻ��5���������⣬Ӧ������C2H5ONa��CH2BrCH2CH2Br��Ȼ����������������Ӧ������Ŀ���
����⣺��1��A�����������ӳɷ�Ӧ����1��3-��������A�к��в����ͼ�������������CO�����������ӳɷ�Ӧ����A��A�к���-CHO����ϩȩת��ΪA��Ӧ����ˮ�����ӳɷ�Ӧ����AΪHOCH2CH2CHO�������ǻ���ȩ����
�ʴ�Ϊ���ǻ���ȩ����
��2����ɫ��ѧ��Ҫ�ɳ�����չ�����ǿɳ�������Դ�������⣬·��1�Կ�������Դ����Ϊԭ�ϣ�·��2��3��ԭ��Ϊʯ�Ͳ�Ʒ����ʯ���Dz���������Դ����·��1����з�չǰ����
�ʴ�Ϊ��1��·��1�Կ�������Դ����Ϊԭ�ϣ�·��2��3��ԭ��Ϊʯ�Ͳ�Ʒ����ʯ���Dz���������Դ��
��3��1��3-��������Ա�������ɷ������۷�Ӧ����Ӧ�ķ���ʽΪ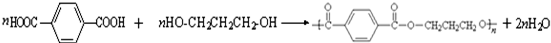 ��
��
�ʴ�Ϊ��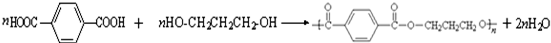 ��
��
��4��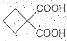 ��ͬ���칹�����Ϊ����-OH�����ڴ������Ժ���-CHO������ȩ������Ϊ���࣮
��ͬ���칹�����Ϊ����-OH�����ڴ������Ժ���-CHO������ȩ������Ϊ���࣮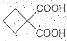 ���Ͷ�Ϊ3�����ຬ�б������䲻���Ͷ�����Ϊ4������ͬ���칹�岻����Ϊ���࣬
���Ͷ�Ϊ3�����ຬ�б������䲻���Ͷ�����Ϊ4������ͬ���칹�岻����Ϊ���࣬
�ʴ�Ϊ��b��
��5���������⣬Ӧ������C2H5ONa��CH2BrCH2CH2Br��Ȼ����������������Ӧ������Ŀ����ɷֱ�����
2CH3CH2OH+2Na��2CH3CH2ONa+H2����HO-CH2CH2CH2-OH+2HBr��BrCH2CH2CH2Br+2H2O��
�ʴ�Ϊ��2CH3CH2OH+2Na��2CH3CH2ONa+H2����HO-CH2CH2CH2-OH+2HBr��BrCH2CH2CH2Br+2H2O��
�ʴ�Ϊ���ǻ���ȩ����
��2����ɫ��ѧ��Ҫ�ɳ�����չ�����ǿɳ�������Դ�������⣬·��1�Կ�������Դ����Ϊԭ�ϣ�·��2��3��ԭ��Ϊʯ�Ͳ�Ʒ����ʯ���Dz���������Դ����·��1����з�չǰ����
�ʴ�Ϊ��1��·��1�Կ�������Դ����Ϊԭ�ϣ�·��2��3��ԭ��Ϊʯ�Ͳ�Ʒ����ʯ���Dz���������Դ��
��3��1��3-��������Ա�������ɷ������۷�Ӧ����Ӧ�ķ���ʽΪ
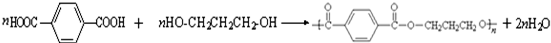 ��
���ʴ�Ϊ��
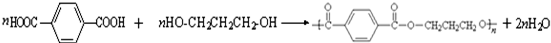 ��
����4��
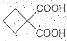 ��ͬ���칹�����Ϊ����-OH�����ڴ������Ժ���-CHO������ȩ������Ϊ���࣮
��ͬ���칹�����Ϊ����-OH�����ڴ������Ժ���-CHO������ȩ������Ϊ���࣮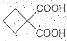 ���Ͷ�Ϊ3�����ຬ�б������䲻���Ͷ�����Ϊ4������ͬ���칹�岻����Ϊ���࣬
���Ͷ�Ϊ3�����ຬ�б������䲻���Ͷ�����Ϊ4������ͬ���칹�岻����Ϊ���࣬�ʴ�Ϊ��b��
��5���������⣬Ӧ������C2H5ONa��CH2BrCH2CH2Br��Ȼ����������������Ӧ������Ŀ����ɷֱ�����
2CH3CH2OH+2Na��2CH3CH2ONa+H2����HO-CH2CH2CH2-OH+2HBr��BrCH2CH2CH2Br+2H2O��
�ʴ�Ϊ��2CH3CH2OH+2Na��2CH3CH2ONa+H2����HO-CH2CH2CH2-OH+2HBr��BrCH2CH2CH2Br+2H2O��
���������⿼���л���ĺϳɣ���Ŀ�Ѷ��еȣ���5��Ϊ�״��㣬������Ŀ�������Ϣ�ǹؼ��������Ŀ��ʱ��ע����չ����ŵĽṹ�����ʣ����ݹ����ŵı仯���з�����

��ϰ��ϵ�д�
�����Ŀ
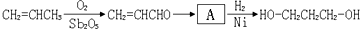

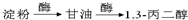
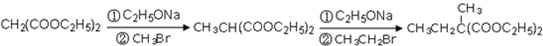
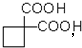 ������Ƴ������ķ�Ӧ����ͼ
������Ƴ������ķ�Ӧ����ͼ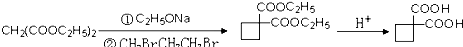
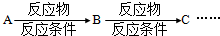
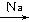
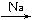

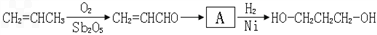
 ��Ϊԭ�Ͽ��Ժϳɾ���PTT��д���仯ѧ����ʽ ______________��
��Ϊԭ�Ͽ��Ժϳɾ���PTT��д���仯ѧ����ʽ ______________��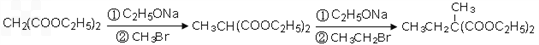
 ����ת��Ϊ
����ת��Ϊ 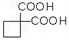 ��
��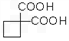 ��ͬ���칹�岻��������__________��
��ͬ���칹�岻��������__________�� 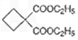 �������Ⱥϳ���Щ���ʣ����úϳɸ����ʵĻ�ѧ����ʽ�ش� __________��
�������Ⱥϳ���Щ���ʣ����úϳɸ����ʵĻ�ѧ����ʽ�ش� __________��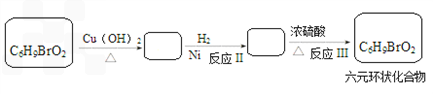
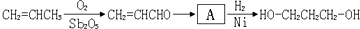

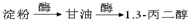
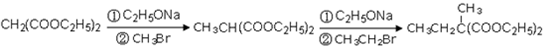
 ������Ƴ������ķ�Ӧ����ͼ______
������Ƴ������ķ�Ӧ����ͼ______