��Ŀ����
������ˮ�п��ܴ��ڵ���ƽ�⡢�ε�ˮ��ƽ��ͳ������ܽ�ƽ�⣬���Ƕ��ɿ�����ѧƽ�⡣�������ѧ֪ʶ�Ļش�
��1��Ũ��Ϊ0.1mol/L��8����Һ����HNO3 ��H2SO4 ��HCOOH ��Ba(OH��2 ��NaOH ��CH3COONa ��KCl ��NH4Cl ��ҺpHֵ��С�����˳����(��д��ţ�____________��
��2��BΪ0.1mol•L-1NaHCO3��Һ��ʵ����NaHCO3��Һ��pH��7���������ӷ���ʽ��ʾ��ԭ��___________________��
��3����0.10 mol•L-1����ͭ��Һ�м�����������ϡ��Һ��ֽ�����dz��ɫ������ͭ�������ɣ�����Һ��pH=8ʱ��c(Cu2+��=___________(��֪Ksp[Cu(OH�� 2]=2.2x10-20��������0.10 mol•L-1����ͭ��Һ��ͨ�����H2S���壬ʹCu2+��ȫ����ΪCuS����ʱ��Һ�е�H+Ũ����________��
��ϰ��ϵ�д�
�����Ŀ


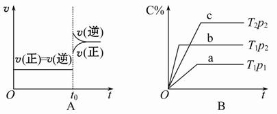
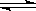 2NH3(g)��t0ʱ�̳�����һ������NH3��ƽ�������ƶ�
2NH3(g)��t0ʱ�̳�����һ������NH3��ƽ�������ƶ� 2SO3(g)�����д�ʩ��ʹ��Ӧ���л���Ӱٷ�������ѧ��Ӧ���ʺͻ�ѧƽ�ⳣ�����仯����
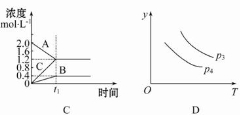
2SO3(g)�����д�ʩ��ʹ��Ӧ���л���Ӱٷ�������ѧ��Ӧ���ʺͻ�ѧƽ�ⳣ�����仯���� xC��g���ڲ�ͬ�¶ȼ�ѹǿ��p1��p2�������·�Ӧ
xC��g���ڲ�ͬ�¶ȼ�ѹǿ��p1��p2�������·�Ӧ ��A��ת���ʵı仯�����ͼ��ʾ�������ж���ȷ���ǣ� ��
��A��ת���ʵı仯�����ͼ��ʾ�������ж���ȷ���ǣ� ��