��Ŀ����
����Ŀ��A��B��C��D���Ƕ�����Ԫ�أ�ԭ�Ӱ뾶��D��C��A��B����֪A��Bͬ���ڣ�A��Cͬ���壬Cԭ�Ӻ��ڵ�����������A��Bԭ�Ӻ��ڵ�������֮�ͣ�Cԭ��������������Dԭ��������������3������ش��������⣺
(1)AԪ�������ڱ��е�λ�ã�____��B��D��Ԫ���γɻ�����Ļ�ѧʽ��_________��
(2)Ԫ��B��C��D�ļ����Ӱ뾶�Ĵ�С˳��____________(�����ӷ��Żش�)��
(3)C��D��Ԫ������������Ӧˮ����ļ���ǿ����ϵ��������������������(�û�ѧʽ��ʾ)��________
(4)B��C�γɵĻ�������DԪ������������Ӧˮ�������Һ�з�Ӧ�Ļ�ѧ����ʽ��____________��
���𰸡��ڶ����ڢ�A�� Na2O��Na2O2 O2->Na+>Al3+ NaOH>Al(OH)3 Al2O3+2NaOH=2NaAlO2+H2O
��������
A��B��C��D���Ƕ�����Ԫ�أ�ԭ�Ӱ뾶D��C��A��B����A��Bͬ���ڣ�A��Cͬ���壬��֪A��B��C��D�����ڱ��еĴ������λ��Ϊ��![]() ��C��ԭ�Ӻ��ڵ�����������A��Bԭ�Ӻ��ڵ�������֮�ͣ�C������=A��������+8����BΪ8��Ԫ��������A��CΪ����Ԫ�أ���C����������ΪD��3������D����������ֻ��Ϊ1����DΪ�ƣ�CΪAl��AΪ�𣬾ݴ˷������
��C��ԭ�Ӻ��ڵ�����������A��Bԭ�Ӻ��ڵ�������֮�ͣ�C������=A��������+8����BΪ8��Ԫ��������A��CΪ����Ԫ�أ���C����������ΪD��3������D����������ֻ��Ϊ1����DΪ�ƣ�CΪAl��AΪ�𣬾ݴ˷������
��������������AΪBԪ�أ�BΪO��CΪAl��DΪNaԪ�ء�
(1)AΪBԪ�أ�ԭ������Ϊ5��λ�����ڱ��еڶ����ڢ�A�壻B��D��Ԫ���γɵĻ�����ΪNa2O��Na2O2���ʴ�Ϊ���ڶ����ڢ�A�壻Na2O��Na2O2��
(2)���Ӳ�ṹ��ͬ�����ӣ��˵����Խ�����Ӱ뾶ԽС�����ӵĵ��Ӳ�Խ�࣬���Ӱ뾶Խ�������Ӱ뾶��СΪ��O2-��Na+��Al3+���ʴ�Ϊ��O2-��Na+��Al3+��
(3)������Na��Al������������Ӧˮ����ļ���ǿ��Ϊ��NaOH��Al(OH)3���ʴ�Ϊ��NaOH��Al(OH)3��
(4)B��C�γɵĻ�����Ϊ��������DԪ������������Ӧˮ����ΪNaOH�����߷�Ӧ�Ļ�ѧ����ʽ��Al2O3+2NaOH�T2NaAlO2+H2O���ʴ�Ϊ��Al2O3+2NaOH�T2NaAlO2+H2O��

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�����Ŀ��ijʵ��С�����ݷ�Ӧ![]() �����ͼ1ԭ��أ�̽��pH��AsO4 3-�����Ե�Ӱ�졣��õ�ѹ��pH�Ĺ�ϵ��ͼ2�������й���������ȷ����( )
�����ͼ1ԭ��أ�̽��pH��AsO4 3-�����Ե�Ӱ�졣��õ�ѹ��pH�Ĺ�ϵ��ͼ2�������й���������ȷ����( )
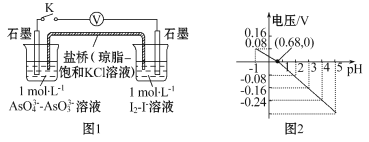
A������pH���Ըı䷴Ӧ�ķ��� |
B��pH=0.68ʱ,��Ӧ����ƽ��״̬ |
C��pH=5ʱ, �����缫��ӦʽΪ2I-��2e -= I2 |
D��pH��0.68ʱ,������I2>AsO43- |
����Ŀ���������Ǻϳ�������66����Ҫԭ��֮һ��ʵ���Һϳɼ������ԭ�����й��������£�
3![]() ��8HNO3��3
��8HNO3��3![]() ��8NO����7H2O
��8NO����7H2O
���� | ��Է������� | �ܶȣ�20�棩 | �۵� | �е� | �ܽ��� |
������ | 100 | 0.962 g/cm3 | 25.9�� | 160.8�� | 20��ʱ����ˮ���ܽ��Ϊ3.6g���ɻ������Ҵ����� |
������ | 146 | 1.360 g/cm3 | 152�� | 337.5�� | ��ˮ�е��ܽ�ȣ�15��ʱ1.44g��25��ʱ2.3g���������Ҵ��������ڱ� |
���������ͼװ�õ�������ƿ�м���16 mL 50%�����ᣨ�������ܶ�Ϊ1.310 g/cm3�����ټ���1��2����ʯ����Һ©����ʢ����5.4 mL��������

�����ˮԡ����������ƿ��50�����ң���ȥˮԡ�������μ�5��6�λ�������ҡ��������ƿ���۲쵽�к���ɫ����ų�ʱ�������μ�ʣ�µĻ�������ά�ַ�Ӧ�¶���60�桫65��֮�䡣
�����������ȫ��������������80�桫90��ˮԡ����Լ10 min��ע������¶ȣ���ֱ������ɫ��������Ϊֹ��
����������Ƚ���ӦҺ�����ձ��У������ˮԡ����ȴ�������������ˡ�ϴ�ӡ�������ء���ش��������⣺
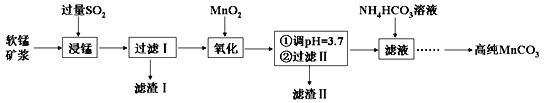
��1��װ��b������Ϊ__________��ʹ��ʱҪ��_________�������Ͽ��������¿�����ͨ����ˮ����Һ©����ϸ֧��a��������________________��
��2��ʵ���У��Ƚ��¶�����������50�����ң�������������60�桫65��֮�䣬��������80�桫90�棬Ŀ����____________________��
��3����ʵ�����õ�50%���������ʵ���Ũ��Ϊ____________��ʵ���У����������������Ҫ�ɷ�ΪNO��NO2��������NaOH��Һ�����գ�����Ҫ��ӦΪNO+NO2+2NaOH == 2NaNO2+H2O������NaOH��Һ������Na2CO3��Һ���������ģ��������Ӧ��д��Na2CO3��Һ���յķ���ʽ��______________________________________��
��4��Ϊ�˳�ȥ���ܵ����ʺͼ��ٲ�Ʒ��ʧ���ɷֱ��ñ�ˮ��______ϴ�Ӿ��塣
��5��ͨ�������õ�����7.00 g����ʵ�����Ϊ__________(��ȷ��0.1��)��
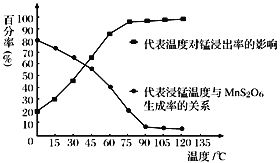
����Ŀ���Է���м(����������)�Ʊ��������(K2FeO4)����������ͼ��ʾ��
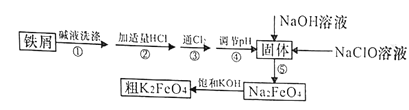
��֪��25��ʱ��һЩ�����������↑ʼ��������ȫ����ʱ��pH���±���ʾ��
M(OH)m | PH | |
��ʼ���� | ������ȫ | |
Fe (OH)3 | 2.53 | 2.94 |
Ni(OH)2 | 7.60 | 9.75 |
(1)K2FeO4����Ԫ�صĻ��ϼ�Ϊ________________��
(2)����Һϴ������Ŀ���dz�ȥ��м��������ۣ�ʵ��һ��ѡ��Na2CO3��Һ���ۣ�ѡ��Na2CO3��Һ���۵�ԭ����____________________________(�����ӷ���ʽ��ʾ)��
(3)����۷�����Ӧ�����ӷ���ʽΪ___________________��
(4)������ǽ�Fe(OH)3��������ΪNa2FeO4��ͬʱNaClOת��ΪNaCl��������1mol Na2FeO4����NaClO������Ϊ______g������ܵ���pH�ķ�Χ��_______��
(5)�õζ����ⶨ���ƴ�K2FeO4�Ĵ���(������KI����Ӧ)��ȡ0.220g��K2FeO4��Ʒ���������������ữ��KI��Һ����ַ�Ӧ����0.200mol��L��1Na2S2O3����Һ�ζ����ɵ�I2���ζ����ı���Һ�����Ϊ20.00mL���漰�ķ�Ӧ�У�FeO42����4I����8H����Fe2����2I2��4H2O��2S2O32����I2��S4O62����2I����
�ٵζ�ʱѡ�õ�ָʾ��Ϊ______���ζ��յ������Ϊ_____________��
�ڴ�K2FeO4�Ĵ���Ϊ_____________��