��Ŀ����
[��ѧ�����ʽṹ������]��13�֣�
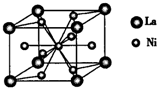
��1��Ԫ�صĵ�һ�����ܣ�Al��Si���>����<������
��2����̬M��+�ĺ�������Ų�ʽΪ ��
��3�����飨SinH2n+2���ķе�������Է��������ı仯��ϵ��ͼ��ʾ���������ֱ仯��ϵ��ԭ���� ��
��4����ɰ�Ǻ��ᾧˮ���������ƣ���������Xm-����B��O��H����Ԫ�أ������ģ����ͼ��ʾ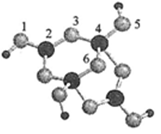
����Xm-�У���ԭ�ӹ�����ӻ������� ����λ�������� ԭ��֮�䣨��ԭ�ӵ����ֱ�ţ���m= �������֣���
����ɰ������Na+��Xm-��H2O���ɣ�����֮����������� ������ţ���
A.���Ӽ� B.���ۼ� C.������ D.���»��� E.���
��1���� ��2��1s22s22p63s23p63d5��[Ar]3d5�� ��3���������Է�������Խ���Ӽ�������Խǿ ��4����sp2��sp3 4��5����5,4�� 2 ��ADE
����

��ϰ��ϵ�д�
�����Ŀ
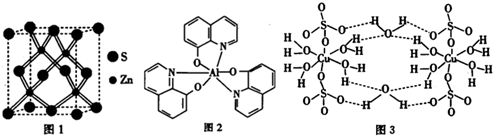
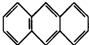 ��ƽ��ṹ������
��ƽ��ṹ������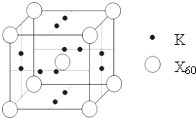 ��2011?����ģ�⣩����ѧ--���ʽṹ�����ʡ�
��2011?����ģ�⣩����ѧ--���ʽṹ�����ʡ�