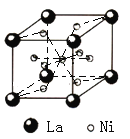
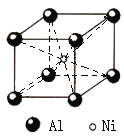
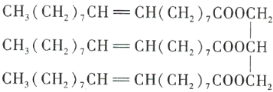��Ŀ����
����Ŀ������һ�����ʵ���Ũ�ȵ���Һ��һ����Ҫ�Ķ���ʵ�飬�����й�˵����ȷ����
A.����ƿ������ˮϴ�����辭����������������Һ
B.��Na2CO310H2O��������Na2CO3��Һʱ��Na2CO3������ʧȥ���ֽᾧˮ���øþ�������Na2CO3��Һ�����ʵ���Ũ��ƫ��
C.����NaOH��Һʱ��NaOH�����к���Na2O���ʣ�������Һ���ʵ���Ũ��ƫС
D.����ʱ��Ϊ��ֹҺ�ηɽ�����ͷ�ιܽ�������ƿ�ڱ�
���𰸡�B
��������
A������ƿ������ˮϴ������Ҫ�������ʱ����Ҫ��ˮ�����ղ�Ӱ��������Һ��Ũ�ȣ���A����
B����Na2CO310H2O��������Na2CO3��Һ��Na2CO3������ʧȥ���ֽᾧˮ���ᵼ��̼�������ʵ���������c=![]() ����nֵ�����øþ�������Na2CO3��Һ�����ʵ���Ũ��ƫ��B��ȷ��
����nֵ�����øþ�������Na2CO3��Һ�����ʵ���Ũ��ƫ��B��ȷ��
C������NaOH��Һʱ��NaOH�����к���Na2O���ʣ������ƺ�ˮ��Ӧ����NaOH��������ͬ���ʵ�����NaOH����������Ҫ������С�����Իᵼ��NaOH�����ʵ���ƫ����c=![]() ����nֵ����������ҺŨ��ƫ��C����
����nֵ����������ҺŨ��ƫ��C����
D������ʱ����ͷ�ι�Ҫ�������ţ����ܽ�������ƿ�ڱڣ�������Ⱦ����D����
��ΪB��

����Ŀ����֪��������������������»ᷢ����Ӧ��![]() ���±��е�������Һ��ϣ����ֻ��ǵ��Ⱥ�˳���ǣ� ��
���±��е�������Һ��ϣ����ֻ��ǵ��Ⱥ�˳���ǣ� ��
��� | ������Һ���¶� |
| ϡ����������Ũ�� |
�� | 15�� | 10mL0.1mol/L | 50mL0.05mol/L |
�� | 15�� | 10mL0.05mol/L | 10mL0.1mol/L |
�� | 25�� | 10mL0.05mol/L | 10mL0.1mol/L |
�� | 25�� | 10mL0.1mol/L | 30mL0.07mol/L |
A.�ܢ٢ڢ�B.�ۢܢڢ�
C.�ܢۢڢ�D.�ܢۢ٢�