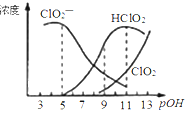
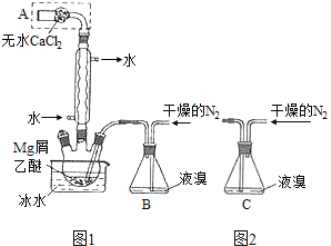
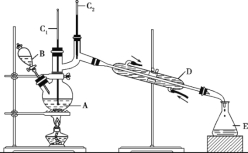
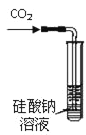
��Ŀ����
����Ŀ��ŨH2SO4 �ܶ�1.84g/mL�����ʵ���Ũ��Ϊ18.4mol/L����������Ϊ98%��ȡ10mLŨH2SO4��a mLˮ��ϵ����ʵ���Ũ��ΪC mol/L����������Ϊb%�����������ȷ���ǣ� ��
��1����C=9.2��a��10 b��49%
��2����C=9.2��a��10 b��49%
��3����b=49��a=18.4 C��9.2
��4����b=49��a=18.4 C��9.2��
A����1����3�� B����1����4�� C����2����3�� D����2����4��
���𰸡�A
��������
�����������C=9.2������ϡ�Ͷ��ɣ���֪ϡ�ͺ���Һ�����Ϊ10mL��![]() =20mL�������Ũ��Խ���ܶ�Խ��ϡ�ͺ���Һ������С��ԭŨ����������2����ϡ����������������䣬��ϡ�ͺ������������������49%��ϡ����������������������䣬��10mL��1.84g/mL��98g%����10mL��1.84g/mL+amL��1g/mL����49%�����a��18.4����b=49��ϡ����������������������䣬��10mL��1.84g/mL��98g%=��10mL��1.84g/mL+amL��1g/mL����49%�����a=18.4����ϡ�ͺ���ܶ�Ϊdg/mL����d��1.84��ϡ��������ʵ���Ũ��C=
=20mL�������Ũ��Խ���ܶ�Խ��ϡ�ͺ���Һ������С��ԭŨ����������2����ϡ����������������䣬��ϡ�ͺ������������������49%��ϡ����������������������䣬��10mL��1.84g/mL��98g%����10mL��1.84g/mL+amL��1g/mL����49%�����a��18.4����b=49��ϡ����������������������䣬��10mL��1.84g/mL��98g%=��10mL��1.84g/mL+amL��1g/mL����49%�����a=18.4����ϡ�ͺ���ܶ�Ϊdg/mL����d��1.84��ϡ��������ʵ���Ũ��C=![]() mol/L��9.2mol/L����A��ȷ��
mol/L��9.2mol/L����A��ȷ��

 ���ɿ��õ�Ԫ������ĩר����100��ϵ�д�
���ɿ��õ�Ԫ������ĩר����100��ϵ�д�