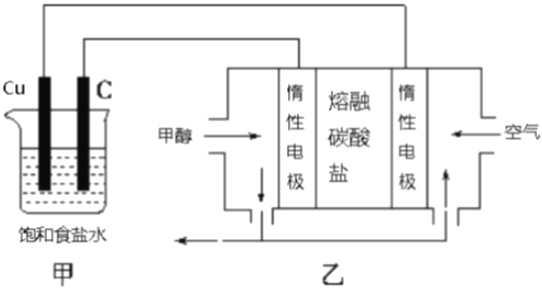
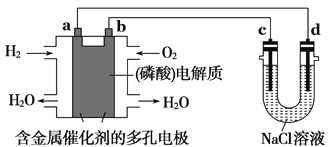
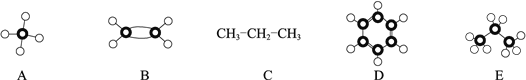��Ŀ����
����Ŀ����ú��ʯ���п�������������ԭ��A��B��A��һ�ֹ�ʵ����������IJ�����������һ�����ҵ�ʯ�ͻ�����չˮƽ��B��һ�ֱ�ˮ�����״Һ�壬B����̼������Ԫ����ɣ�̼Ԫ������Ԫ�ص�������Ϊ12��1��B����Է�������Ϊ78���ش��������⣺
��1��A�ķ���ʽ_______��B�Ľṹ��ʽ_______��
��2��A��ʹ������Ȼ�̼��Һ��ɫ���仯ѧ��Ӧ����ʽΪ____________________________��
��3���ڵ�ˮ�м���B���ú������________________��
��4��B��Ũ�����Ũ������50��60�淴Ӧ�Ļ�ѧ��Ӧ����ʽ��________________________����Ӧ����_______��
��5��B��ͬϵ�����ʽΪC8H10������ʹ���Ը��������ɫ������ʹ��ˮ��ɫ��������ͬ���칹����____________�֣�д��ֻ��һ��ȡ������C8H10�Ľṹ��ʽ________________��
���𰸡� C2H4 ![]() CH2=CH2+Br2��CH2Br-CH2Br �²���ɫ���ϲ��Ϻ�ɫ
CH2=CH2+Br2��CH2Br-CH2Br �²���ɫ���ϲ��Ϻ�ɫ ![]() ȡ����Ӧ 4��
ȡ����Ӧ 4�� 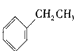
��������A��һ�ֹ�ʵ����������IJ�����������һ�����ҵ�ʯ�ͻ�����չˮƽ����AΪCH2=CH2��B��һ�ֱ�ˮ�����״Һ�壬B����̼������Ԫ����ɣ�̼Ԫ������Ԫ�ص�������Ϊ12��1����ԭ�Ӹ���֮��Ϊ1��1������CnH2n-6��B����Է�������Ϊ78����12n+2n-6=78�����n=6������BΪ����������ϩ�ͱ����������ʺͻ�ѧ����ȥ�жϷ�����������⣬����ͬ���칹���д������C8H10��ͬ���칹����Ŀ��
��1��������ɷ�����֪��A����ϩ�������ʽ�ǣ�C2H4��B�DZ�����ṹ��ʽ��![]() ����ȷ�𰸣�C2H4��
����ȷ�𰸣�C2H4��![]() ��
��
��2��CH2=CH2ʹ��ˮ��ɫ�������ӳɷ�Ӧ���÷�ӦΪCH2=CH2+Br2��CH2Br-CH2Br���ʴ�Ϊ��CH2=CH2+Br2��CH2Br-CH2Br��
��3����ˮ�м��뱽������ȡ�������ܶȱ�ˮ��С�������ϲ㣬�۲쵽�²���ɫ���ϲ��Ϻ�ɫ����ȷ��Ϊ���²���ɫ���ϲ��Ϻ�ɫ��
��4��B��ŨH2SO4��ŨHNO3��50-60����Ӧ�Ļ�ѧ��Ӧ����C6H6+HO-NO2![]() C6H5NO2+H2O������ȡ����Ӧ����ȷ��Ϊ��C6H6+HO-NO2
C6H5NO2+H2O������ȡ����Ӧ����ȷ��Ϊ��C6H6+HO-NO2![]() C6H5NO2+H2O��ȡ����Ӧ��
C6H5NO2+H2O��ȡ����Ӧ��
��5��C8H10�DZ���ͬϵ�����ʹ���Ը��������ɫ������ʹ��ˮ��ɫ����֧��Ϊ�һ�ʱ����ṹֻ��һ��![]() ����֧����2����ʱ����ṹ��ʽ�����֣�
����֧����2����ʱ����ṹ��ʽ�����֣�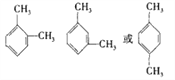 ������C8H10�ı���ͬϵ���ͬ���칹�干��4������ȷ�𰸣�4��
������C8H10�ı���ͬϵ���ͬ���칹�干��4������ȷ�𰸣�4�� ��
��

����Ŀ����ȥ���������е����ʣ������Լ��ͷ�����ȷ���ǣ� ��
���� | ���� | �����������Լ��ͷ��� | |
A | FeCl3 | FeCl2 | ����������Fe�ۣ����� |
B | CO2 | HCl | ͨ�뱥��Na2CO3��Һ��ϴ�� |
C | SO2 | H2O | ͨ��Ũ���ᣬϴ�� |
D | Mg | Cu | ����Ũ���ᣬ���� |
A.AB.BC.CD.D