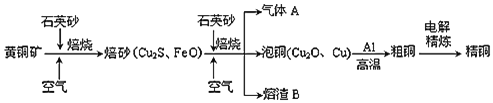
ЬтФПФкШн
ЁОЬтФПЁПЯђЪЂгаСђЫсЭЫЎШмвКЕФЪдЙмРяМгШыАБЫЎЃЌЪзЯШаЮГЩФбШмЮяЃЌМЬајМгАБЫЎЃЌФбШмЮяШмНтЃЌЕУЕНЩюРЖЩЋЕФЭИУїШмвКЃЛШєМгШыМЋадНЯаЁЕФШмМС(ШчввДМ)ЃЌНЋЮіГіЩюРЖЩЋЕФОЇЬхЁЃ
Ђё.дкЛЏбЇЪЕбщКЭПЦбЇбаОПжаЃЌЫЎЪЧвЛжжзюГЃгУЕФШмМСЁЃЫЎЪЧЩњУќжЎдДЃЌЫќгыЮвУЧЕФЩњЛюУмЧаЯрЙиЁЃ
(1)аДГіH2OЗжзгЕФЕчзгЪНЃК________ЁЃ
(2)ЫЎЗжзгдкЬиЖЈЬѕМўЯТШнвзЕУЕНвЛИіHЃЋЃЌаЮГЩЫЎКЯЧтРызг(H3OЃЋ)ЁЃЯТСаЖдЩЯЪіЙ§ГЬЕФУшЪіВЛКЯРэЕФЪЧ________ЁЃ
AЃЎбѕдзгЕФдгЛЏРраЭЗЂЩњСЫИФБф
BЃЎЮЂСЃЕФаЮзДЗЂЩњСЫИФБф
CЃЎЮЂСЃЕФЛЏбЇаджЪЗЂЩњСЫИФБф
DЃЎЮЂСЃжаЕФМќНЧЗЂЩњСЫИФБф
Ђђ.ЕЈЗЏОЇЬхЪЧХфжЦВЈЖћЖрвКЕФжївЊдСЯЃЌВЈЖћЖрвКЪЧвЛжжБЃЛЄадЩБОњМСЃЌЙуЗКгІгУгкЪїФОЁЂЛЈЛмЩЯЁЃ
(3)аДГіЭдзгМлЕчзгВуЕФЕчзгХХВМЪНЃК_____ЃЌгыЭЭЌвЛжмЦкИБзхдЊЫиЕФЛљЬЌдзгжазюЭтВуЕчзгЪ§гыЭдзгЯрЭЌЕФдЊЫига________(ЬюдЊЫиЗћКХ)ЁЃ
(4)ЪЕбщЪБаЮГЩЕФЩюРЖЩЋШмвКжаЕФбєРызгФкДцдкЕФШЋВПЛЏбЇМќРраЭга________ЁЃ
(5)ЪЕбщЙ§ГЬжаМгШыC2H5OHКѓПЩЙлВьЕНЮіГіЩюРЖЩЋCu(NH3)4SO4ЁЄH2OОЇЬхЁЃЪЕбщжаЫљМгC2H5OHЕФзїгУЪЧ_________________________ ЁЃ
ЁОД№АИЁП![]() A 3d104s1 Cr ЙВМлМќЁЂХфЮЛМќ НЕЕЭCu(NH3)4SO4ЁЄH2OЕФШмНтЖШ
A 3d104s1 Cr ЙВМлМќЁЂХфЮЛМќ НЕЕЭCu(NH3)4SO4ЁЄH2OЕФШмНтЖШ
ЁОНтЮіЁП
Ђё.ЃЈ1ЃЉЫЎЮЊЙВМлЛЏКЯЮяЃЛ
ЃЈ2ЃЉA. ИљОнМлВуЕчзгЖдЛЅГтРэТлШЗЖЈжааФдзгдгЛЏРраЭЃЌМлВуЕчзгЖдИіЪ§=![]() МќИіЪ§+ЙТЕчзгЖдИіЪ§ЃЌЫЎжабѕЕФдгЛЏЗНЪНЮЊsp3ЃЌH3OЃЋжаOЕФдгЛЏЗНЪНЮЊsp3ЃЛ
МќИіЪ§+ЙТЕчзгЖдИіЪ§ЃЌЫЎжабѕЕФдгЛЏЗНЪНЮЊsp3ЃЌH3OЃЋжаOЕФдгЛЏЗНЪНЮЊsp3ЃЛ
B. ЫЎЮЊVаЮНсЙЙЃЌH3OЃЋЮЊШ§НЧзЖаЮЃЛ
C. ЗжзгКЭРызгЕФЛЏбЇаджЪВЛЭЌЃЛ
D. ИљОнЮЂСЃЕФПеМфЙЙаЭХаЖЯЃЛ
Ђђ.ЃЈ3ЃЉCuЕФдзгађЪ§ЮЊ29ЃЌЮЊЕкЫФжмЦкЕкIBзхдЊЫиЃЌзюЭтВуЕчзгЮЊ4s1ЃЛ
ЃЈ4ЃЉЕЈЗЏШмвКгызуСПАБЫЎаЮГЩЕФЩюРЖЩЋШмвКжаЕФбєРызгЮЊЫФАБКЯЭТчРызгЃЛ
ЃЈ5ЃЉДгНЕЕЭЮяжЪЕФШмНтЖШЕФНЧЖШНјааЗжЮіЁЃ
Ђё.ЃЈ1ЃЉЫЎЮЊЙВМлЛЏКЯЮяЃЌЕчзгЪНЮЊЃК![]() ЃЛ
ЃЛ
ЃЈ2ЃЉA. ЫЎЗжзгжаOдзгМлВуЕчзгЖдИіЪ§=2+![]() ЁС(6-2ЁС1)=4ЃЌВЩгУsp3дгЛЏЃЌH3OЃЋжаOдзгМлВуЕчзгЖдИіЪ§=3+
ЁС(6-2ЁС1)=4ЃЌВЩгУsp3дгЛЏЃЌH3OЃЋжаOдзгМлВуЕчзгЖдИіЪ§=3+![]() ЁС(6-1-3ЁС1)=4ЃЌВЩгУsp3дгЛЏЃЌбѕдзгЕФдгЛЏРраЭУЛгаИФБфЃЌAЯюДэЮѓЃЌЗћКЯЬтвтЃЛ
ЁС(6-1-3ЁС1)=4ЃЌВЩгУsp3дгЛЏЃЌбѕдзгЕФдгЛЏРраЭУЛгаИФБфЃЌAЯюДэЮѓЃЌЗћКЯЬтвтЃЛ
B. ЫЎЗжзгжаOдзгКЌга2ИіЙТЕчзгЖдЃЌВЩгУsp3дгЛЏЃЌЃЌПеМфЙЙаЭЮЊVаЮНсЙЙЃЌH3OЃЋЮЊШ§НЧзЖаЮЃЌЮЂСЃЕФаЮзДЗЂЩњСЫБфЛЏЃЌBЯюе§ШЗЃЌВЛЗћКЯЬтвтЃЛ
C. H2OЮЊжаадЗжзгЃЌH3OЃЋЮЊЫсадЕФбєРызгЃЌЮЂСЃЕФЛЏбЇаджЪЗЂЩњСЫБфЛЏЃЌCЯюе§ШЗЃЌВЛЗћКЯЬтвтЃЛ
D. ЫЎЗжзгЮЊVаЮНсЙЙЃЌH3OЃЋЮЊШ§НЧзЖаЮЃЌЮЂСЃжаЕФМќНЧЗЂЩњСЫБфЛЏЃЌDЯюе§ШЗЃЌВЛЗћКЯЬтвтЃЛ
Д№АИбЁAЁЃ
Ђђ.ЃЈ3ЃЉCuЕФдзгађЪ§ЮЊ29ЃЌЮЊЕкЫФжмЦкЕкIBзхдЊЫиЃЌМлВуЕчзгЕФЕчзгХХВМЮЊ3d104s1ЃЌзюЭтВуЮЊ4s1ЃЌЭЌжмЦкЕФИБзхдЊЫиCrЕФМлВуЕчзгХХВМЮЊ3d54s1ЃЛ
ЃЈ4ЃЉЕЈЗЏШмвКгызуСПАБЫЎаЮГЩЕФЩюРЖЩЋШмвКжаЕФбєРызгЮЊЫФАБКЯЭТчРызгЃЌNЁЂHдзгжЎМфвдЙВМлМќНсКЯЃЌCu2+ЬсЙЉПеЙьЕРЃЌNдзгЬсЙЉЙТЖдЕчзгЃЌвдХфЮЛМќНсКЯЃЛ
ЃЈ5ЃЉЪЕбщЙ§ГЬжаМгШыC2H5OHКѓЃЌНЕЕЭСЫCu(NH3)4SO4ЕФШмНтЖШЃЌЮіГіСЫCu(NH3)4SO4ЁЄH2OОЇЬхЁЃ

 ПкЫуаФЫуЫйЫугІгУЬтЯЕСаД№АИ
ПкЫуаФЫуЫйЫугІгУЬтЯЕСаД№АИ ЭЌВНЭиеЙдФЖСЯЕСаД№АИ
ЭЌВНЭиеЙдФЖСЯЕСаД№АИЁОЬтФПЁПЮвЙњЙцЖЈвћгУЫЎжЪСПБъзМБиаыЗћКЯЯТБэЫљЪОвЊЧѓЃК
ЯюФП | pH | Ca2+ЁЂMg2+змХЈЖШ | ЯИОњИіЪ§ |
ЯрЙижЕ | 6.5ЁЋ8.5 | <0.0045molЁЄLЃ1 | <100ИіЁЄmLЃ1 |
ЯТЭМЪЧдДЫЎДІРэГЩздРДЫЎЕФЙЄвеСїГЬЪОвтЭМЁЃ

(1)дДЫЎжаКЌCa2+ЁЂMg2+ЁЂHCO3-ЁЂCl-ЕШЃЌМгШыЪЏЛвКѓЩњГЩCa(OH)2ЃЌНјЖјЗЂЩњШєИЩИДЗжНтЗДгІЃЌдквЛМЖГСНЕГижаЫљЕУГСЕэЕФжївЊГЩЗжЮЊ _____ КЭ_______ (ЛЏбЇЪН)ЁЃ
(2)FeSO4ЁЄ7H2OЪЧГЃгУЕФФ§ОлМСЃЌЫќдкЫЎжазюжеЩњГЩНКзДFe(OH)3ГСЕэЁЃФ§ОлМСГ§ШЅаќИЁЙЬЬхПХСЃЕФЙ§ГЬ______________(ЬюаДБрКХ)ЁЃ
ЂйжЛЪЧЮяРэЙ§ГЬЃЛ ЂкжЛЪЧЛЏбЇЙ§ГЬЃЛ ЂлЪЧЮяРэКЭЛЏбЇЙ§ГЬЁЃ
(3)Cl2ЕФзїгУЪЧ______ ЃЌетжжзїгУЪЧЛљгкCl2КЭЫЎЗДгІЕФВњЮяОпга _______ адЁЃ
ЁОЬтФПЁП10ЁцЪБМгШШNaHCO3БЅКЭШмвКЃЌВтЕУИУШмвКЕФpHЗЂЩњШчЯТБфЛЏ
ЮТЖШ(Ёц) | 10 | 20 | 30 | МгШШжѓЗаКѓРфШДЕН50Ёц |
pH | 8.3 | 8.4 | 8.5 | 8.8 |
(1)МзЭЌбЇШЯЮЊЃЌИУШмвКЕФpHжЕЩ§ИпЕФдвђЪЧHCO3ЃЕФЫЎНтГЬЖШдіДѓЃЌЙЪМюаддіЧПЃЌИУЗДгІЕФРызгЗНГЬЪНЮЊ_________ЁЃ
(2)ввЭЌбЇШЯЮЊЃЌШмвКpHЩ§ИпЕФдвђЪЧNaHCO3ЪмШШЗжНтЃЌЩњГЩСЫNa2CO3ЃЌВЂЭЦЖЯNa2CO3ЕФЫЎНтГЬЖШ_________(ЬюЁАДѓгкЁБЛђЁАаЁгкЁБ)NaHCO3ЁЃ
(3)БћЭЌбЇШЯЮЊМзЁЂввЕФХаЖЯЖМВЛГфЗжЁЃБћШЯЮЊЃК
ЂйжЛвЊдкМгШШжѓЗаЕФШмвКжаМгШызуСПЕФЪдМСXЃЌШєВњЩњГСЕэЃЌдђ_________(ЬюЁАМзЁБЛђЁАввЁБ)ХаЖЯе§ШЗЁЃЪдМСXЪЧ_____________(ЬюађКХ)ЁЃ
a.Ba(OH)2ШмвК b.BaCl2ШмвК c.NaOHШмвК d.ГЮЧхЪЏЛвЫЎ
ЂкНЋМгШШКѓЕФШмвКРфШДЕН10ЁцЃЌШєШмвКЕФpH____________(ЬюЁАИпгкЁБЁЂЁАЕЭгкЁБЛђЁАЕШгкЁБ)8.3ЃЌдђМзХаЖЯе§ШЗЁЃ
(4)ГЃЮТЯТЃЌдкВтЕУpHЖМЕШгк9ЕФNaOHШмвККЭNa2CO3ШмвКжаЃЌгЩЫЎЕчРыГіЕФOHЃХЈЖШЗжБ№ЮЊa mol/LКЭb mol/LЁЃдђaЃКbЃН________ЁЃ
(5)25ЁцЪБЃЌХЈЖШОљЮЊ0.1 mol/LЕФNa2CO3КЭNaHCO3ШмвКЁЃ
ЂйЯТСаХаЖЯВЛе§ШЗЕФЪЧ________(ЬюађКХ)ЁЃ
a.СНжжШмвКЕШЬхЛ§ЛьКЯКѓЃК3c(NaЃЋ)ЃН2[c(HCO3Ѓ)ЃЋc(CO32Ѓ)ЃЋc(H2CO3)]
b.СНжжШмвКжаДцдкЕФСЃзгжжРрВЛЯрЭЌ
c.СНжжШмвКжаc(HЃЋ)ЁЄc(OHЃ)ЃН10Ѓ14
d.СНжжШмвКжаЗжБ№МгШыЩйСПNaOHЙЬЬхЃЌЛжИДЕНдЮТЖШЃЌc(CO32Ѓ)ОљдіДѓ
ЂкЕШЬхЛ§ЛьКЯКѓШмвКжаРызгХЈЖШгЩДѓЕНаЁЕФЫГађЪЧЃК__________ЃЛ
ЂлГЃЮТЯТЃЌЕШЬхЛ§ЛьКЯКѓЕФШмвКжаЕЮМгЯЁбЮЫсжСжаадЪБЃЌШмжЪЕФжївЊГЩЗжгаЃК_______ЁЃ