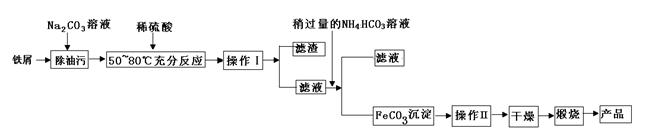
��Ŀ����
�����������壨FeSO4·7H2O����ҽҩ������Ѫ����ij����С��ⶨ�ò�Ѫ������Ԫ�صĺ�����������ò�Ѫ���Ƿ���ʡ�ʵ�鲽�����£�
��ش��������⣺
��1���������Һ�еμ�KSCN��Һ����Һ��Ϊ��ɫ�������Һ�к��� (�����ӷ���)��������Һ�л�����Fe2+�ķ���Ϊ
��ע���Լ�������
��2������ڼ������H2O2��Ŀ���� ��
��3��������з�Ӧ�����ӷ���ʽΪ ��
��4���������һϵ�д����IJ���������������ˡ� �����ա� ��������
��5����ʵ������ģ���ÿƬ��Ѫ������Ԫ�ص�����Ϊ g��
��1��Fe3+ ȡһ������Һ���μ�����KMnO4��Һ��KMnO4��Һ��ɫ
��2����Fe2+ȫ������ΪFe3+
��3��Fe3++3OH��= Fe��OH��3������Fe3++3NH3·H2O = Fe��OH��3+3NH4+��
��4��ϴ�� ��ȴ
��5��0.07a (��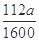 ���仯����ʽ����
���仯����ʽ����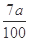 )
)
���������������1��KSCN��Һ��Fe3+��Ӧ��Ѫ��ɫ����Fe3+���ڵ�����������Fe2+�Ļ�ԭ�Լ�������ڣ�ȡһ������Һ���μ�����KMnO4��Һ��KMnO4��Һ��ɫ����2��H2O2�ǰ�Fe2+����ΪFe3+��������Ϊ�˽�Fe2+ȫ������ΪFe3+����3������Һ�м���ǿ����Һ��������������������4��Ϊ�˵õ�����������������Ҫϴ�ӣ����գ���ȴ��õ�����5������Ϊ��������10Ƭ��Ѫ���õ��ģ�ÿƬ��Ѫ��������Ԫ�ص�����Ϊa��160��56��2��10="0.07a" g��
���㣺���黯ѧʵ�鶨�������й����⡣

 �������ͬ������ϵ�д�
�������ͬ������ϵ�д�������������Ҫ�������Σ���ũҵ������ũҩ����Ҫ��С����벡���������������ݼ����ڹ�ҵ������Ⱦɫ����������īˮ��ľ�ķ����ȡ�
��1�����Ƶ��̷���FeSO4��7H2O����dz��ɫ�ģ����ڿ����м��ױ�ɻ�ɫ������ɫ�ļ�ʽ������[Fe(OH)SO4]��д���÷�Ӧ�Ļ�ѧ����ʽ�� ��
��2����֪FeSO4�ڲ�ͬ�����·ֽ�õ����ﲻͬ��������FeO��SO3��Ҳ������Fe2O3��SO3��SO2��
SO3�۵���16.8�棬�е���44.8�档
ij�о���ѧϰС����������װ�ý���ʵ��̽�����ڼ���������FeSO4�ķֽ�����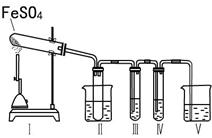
����װ�â�͢����������������Իش��������⣺
�٢�װ���ձ���ˮ���¶�Ӧ������ ��ѡ�0�桢25�桢50�桱����װ�â�������� ��
��װ�â��е��Լ������� ��ѡ����ţ���ͬ���������� ����֤����������к���SO2�� װ�â��е��Լ������� ��
| A��2 mol/LNa2CO3��Һ |
| B��Ʒ����Һ |
| C��0.5 mol/LBaCl2��Һ |
| D��0.5 mol/LBa(NO3)2 |
F. ���۵⻯����Һ
��װ��V���Լ�ΪNaOH��Һ��������Ӧ�����ӷ���ʽΪ ��
��Ϊ�˼���������ɷ֣�ȡ��Ӧ��Ĺ������Թ��У���ϡ�����ܽ⣬��������Һ�ֳ����ݣ�������
��ʵ�飺
| �������� | Ԥ��ʵ������ | Ԥ��ʵ����� |
| ������һ����Һ�м��� �� | | �����к���Fe2O3 |
| ����һ����Һ�еμ�2�λ�ɫK3[Fe(CN)6]��Һ�� | ������ɫ���� | |
������22.8 g FeSO4������ʵ�飬��ȫ�ֽ�õ�11.2 g���壬����Fe2O3����������=
����ȷ��0.1%��
С����ϲ����ѧʵ��Σ�����Ҫѧϰ��̽�������仯����������Ի�ԭ�ԡ���������һ���߽����Ļ�ѧ���á�
��1����ǰ��ʦ����������Ԥϰ��ҵ������һ����ɣ�
������ͬ��̬�����ʸ�дһ��(�������Ԫ�صĻ��ϼ�)��_____��_______�� ________��
��д��һ������֮���ת��(�����ּ�̬)�Ļ�ѧ����ʽ��___________________ ��
��2��ʵ�����ṩ�������Լ���п�������ۡ�0.1 mol��L��1 FeCl3��Һ��0.1 mol��L��1 FeCl2��Һ��KSCN��Һ��������ˮ��̽��Fe2����Fe3���������ԡ���ԭ�ԡ�
����������ԭ��Ӧ���й�ԭ����С��˵Fe2�����л�ԭ�����������ԣ�Ϊ֤ʵ�Լ��ļ��裬�����С��һ�����ʵ�鷽��������ʵ�鲢����ʵ������������б���
| ̽������ | ʵ�鷽�� | ʵ������ |
| ̽��Fe2�����л�ԭ�� | ȡ����0.1 mol��L��1 FeCl2��Һ����������__________��������Һ�м�������__________ | ��Һ���Ѫ��ɫ |
| ̽��Fe2������������ | ȡ����0.1 mol��L��1 FeCl2�� Һ������_________��� ��Ӧ | ��Һ��dz��ɫ����ɫ ��������Ӧ���ӷ���ʽΪ________________ [��Դ:Z|xx|k.Com] |
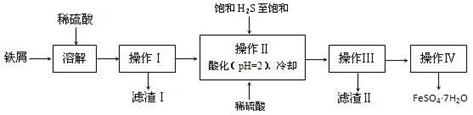
FeCl3���ִ���ҵ������Ӧ�ù㷺��ij��ѧ�о���ѧϰС��ģ�ҵ�����Ʊ���ˮFeCl3�����ø���ƷFeCl3��Һ�����ж���H2S��
I�����������ϵ�֪����ˮFeCl3�ڿ������׳��⣬����������������������Ʊ���ˮ FeCl3��ʵ�鷽����װ��ʾ��ͼ�����ȼ��г�װ����ȥ���������������£�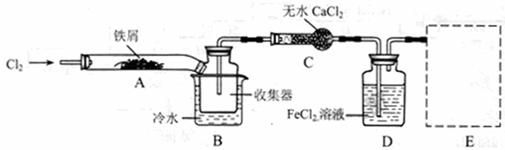
�ټ���װ�õ������ԣ�
��ͨ������Cl2���Ͼ�װ���еĿ�����
���þƾ�������м�·���������Ӧ��ɣ�
�ܡ���
����ϵ��ȴ��ֹͣͨ��Cl2�����ø����H2�Ͼ�Cl2�����ռ����ܷ⡣
��ش��������⣺
��1��װ��A�з�Ӧ�Ļ�ѧ����ʽΪ ��
��2���ڢ۲����Ⱥ����ɵ���״FeCl3�ֽ����ռ��������������ڷ�Ӧ��A�Ҷˡ�Ҫʹ������FeCl3�����ռ������ڢܲ������� ��
��3�����������У�Ϊ��ֹFeCl3��������ȡ�Ĵ�ʩ�У������ţ� ��
��4�����û��װ��C����ƣ��ᵼ�� ��
��5�������ӷ���ʽ��ʾ���߿�E��������װ�ú��Լ������ã� ��
��6����װ��D�еĸ���ƷFeCl3��Һ����H2S���õ�������
��д����Ӧ�����ӷ���ʽ�� ��
��Ӧ�������ռ��������ù�����ȫ����ϡ���ᣬС��ͬѧ��������Һ���������ӵijɷ������ֹ۵㣺��ֻ��Fe3+����ֻ��Fe2+���� ��
Ϊ̽����Һ����ɣ�ʵ�����£�
| ʵ�鲽�� | ʵ������ | ʵ����ۼ���Ӧ���ӷ���ʽ |
| ��ȡ����������Һ���Թ��У���������KSCN��Һ�� | | ˵��������ڲ�����������ٻ�۳����������ӷ���ʽ�� �� |
| ����ȡ����������Һ���Թ��У������������� KMnO4��Һ�� | ��Һ�Ϻ�ɫ��ȥ | ˵���� �� |
�ۺ�����ʵ�������������Һ�к��еĽ����������� ��
��ͼ����ע�����м�������Na2SO3���壬����������Ũ����(�Բ��Ӵ�ֽ��Ϊ)���������й�˵����ȷ����(����) 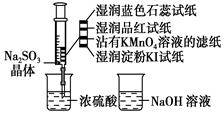
| A����ɫʯ����ֽ�ȱ�����ɫ |
| B��Ʒ����ֽ��մ��KMnO4��Һ��ֽ����ɫ����֤��SO2��Ư���� |
| C��ʪ�����KI��ֽδ����˵��SO2������������I2 |
| D��NaOH��Һ��Ʒ����Һ�������ڳ�ȥʵ���ж����SO2 |
 Fe��SCN��3���з�ӦѸ�١��������Ե��ص㣬�Ǽ���Fe3�����õķ���֮һ��ij��ѧ��ȤС��Ϊ̽��Fe��SCN��3�����ʣ���������ʵ�飺
Fe��SCN��3���з�ӦѸ�١��������Ե��ص㣬�Ǽ���Fe3�����õķ���֮һ��ij��ѧ��ȤС��Ϊ̽��Fe��SCN��3�����ʣ���������ʵ�飺