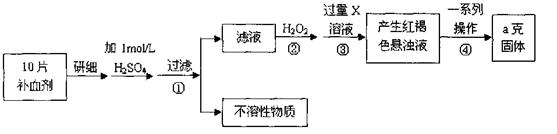
��Ŀ����
��֪���ӷ�Ӧ��Fe3����3SCN�� Fe��SCN��3���з�ӦѸ�١��������Ե��ص㣬�Ǽ���Fe3�����õķ���֮һ��ij��ѧ��ȤС��Ϊ̽��Fe��SCN��3�����ʣ���������ʵ�飺
Fe��SCN��3���з�ӦѸ�١��������Ե��ص㣬�Ǽ���Fe3�����õķ���֮һ��ij��ѧ��ȤС��Ϊ̽��Fe��SCN��3�����ʣ���������ʵ�飺
��ȡ10 mL l mol��L��1FeCl3��Һ���μ�3��4��ŨKSCN��Һ������Һ������ɺ�
ɫ��
��ȡ������ɫ��Һ���μ�����Ũ���ᣬ���ã���Һ��ɫ��ȥ��ͬʱ���������ĺ���ɫ��
������A��
�۽�����������Aͨ�������Ba��OH��2��Һ�У�������ɫ����B��ʣ������C��
����C��ɫ��ζ����ʹȼ�յ�ľ��Ϩ�𣬿��ŷŵ������У�����ı�����ijɷ֡�
�ܹ��ˣ����ɫ����B�еμ�����ϡ���ᣬ������ȫ�ܽ⣬ͬʱ������ʹ����ʯ��ˮ��
���ǵ���ɫ��ζ����D��
��ȡ���з�Ӧ����Һ�������μ�BaCl2��Һ������������ϡ����İ�ɫ����E��
��������ʵ�����ش��������⣺
��1��B�Ļ�ѧʽΪ_________________��E�Ļ�ѧʽΪ___________________��
��2���������A�ijɷ���___________________���ѧʽ����
��3������ȤС��ͬѧ��������ʵ����������ó����ۣ�Fe��SCN��3���л�ԭ�ԣ���ʵ����з�Ӧʱ��������Ԫ����____________����Ԫ�ط��ţ�������Ԫ��C��S��N��ԭ�ӽṹ���ۼ������֪ʶ�ƶϳ�SCN���ĽṹʽΪ_________________��
��4��ʵ����з�Ӧ�����ӷ���ʽΪ___________________________________________��
��5������ȤС��ͬѧ������ʵ���еõ�����������SCN����Ӽ���Fe2��ʱӦע��_________________________________________________________________.
��1��BaCO3(1��); BaSO4(1��)
��2��NO2��CO2��N2��2�֣�
��3��S��N��2�֣���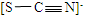 ��1�֣�
��1�֣�
��4��2SCN����22NO3����20H��=2SO42����2CO2����22NO2����N2��10H2O��2�֣�
��5���������������ܹ�����2�֣�
��������������ɢ��еĺ���ɫ�������A��֪A����NO2,�ɢۢܿ�֪BΪBaCO3��CΪN2��DΪCO2����A��NO2��N2��CO2��ɣ��ɢݿ�֪EΪBaSO4��2�� ��A��NO2��N2��CO2��ɣ�3���ɲ��PԪ�صĻ��ϼۿ�֪��������Ԫ����S��N������Ԫ��C��S��N��ԭ�ӽṹ���ۼ������֪ʶ�ƶϳ�SCN���ĽṹʽΪ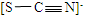 ����4�����������P�����غ�͵���غ㡢�����غ���ƽ����ʽΪ��2SCN����22NO3����20H��=2SO42����2CO2����22NO2����N2��10H2O��5���������֪SCN-Ҳ�ܱ��������������ʼ�������������ܹ���
����4�����������P�����غ�͵���غ㡢�����غ���ƽ����ʽΪ��2SCN����22NO3����20H��=2SO42����2CO2����22NO2����N2��10H2O��5���������֪SCN-Ҳ�ܱ��������������ʼ�������������ܹ���
���㣺����������ԭ��Ӧ�����ʽṹ�����ʵ����ʵ�֪ʶ�㡣

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д��������Ũ�������ܷ����ۻ���ij��ȤС���ͬѧ���ֽ�һ����������Ũ�������ʱ���۲쵽����ȫ�ܽ⣬�������������塣ʵ�������������Լ���0.01 mol/L ����KMnO4��Һ��0.1 mol/L KI��Һ��3��H2O2��Һ��������Һ������ˮ������Э������̽��������Һ������ijɷ֡�
��������롿
��.������Һ�еĽ������ӿ��ܺ���Fe2+��Fe3+�е�һ�ֻ����֣�
��.���������п��ܺ���_________�е�һ�ֻ����֡�
| | ʵ����� | Ԥ������ | ���� |
| ��֤����� | ����٣�ȡ����0.01 mol/L����KMnO4��Һ������������Һ | | |
| ����ڣ�_________ | | ����Fe3�� | |
| ��֤����� | ����������ͨ������װ�� | | ������������ |
��ʵ��̽����

���������ۡ�
��ͬѧ�����������ѡ��KSCN��Һ���������KSCN��H2O2������Һ������ɲ���������̽�����жϸ÷����Ƿ���ȷ���������ۣ�_________��
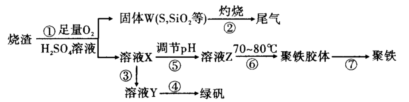
FeCl3���ִ���ҵ������Ӧ�ù㷺��ij��ѧ�о���ѧϰС��ģ�ҵ���������Ʊ���ˮFeCl3�����ø���ƷFeCl3��Һ�����ж���H2S��
���������ϵ�֪����ˮFeCl3�ڿ������׳��⣬����������������������Ʊ���ˮFeCl3��ʵ�鷽����װ��ʾ��ͼ(���ȼ��г�װ����ȥ)�������������£�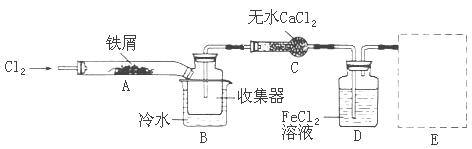
�ټ��װ�õ������ԣ�
��ͨ������Cl2���Ͼ�װ���еĿ�����
���þƾ�������м�·���������Ӧ���
�� ����
����ϵ��ȴ��ֹͣͨ��Cl2�����ø����N2�Ͼ�Cl2�����ռ����ܷ�
��ش��������⣺
��1��д��FeCl3��Һ�����ж���H2S�����ӷ���ʽΪ
��2���ڢ۲����Ⱥ����ɵ���״FeCl3�ֽ����ռ��������������ڷ�Ӧ��A���Ҷˡ�Ҫʹ������FeCl3�����ռ������ڢܲ�������
��3�����������У�Ϊ��ֹFeCl3��������ȡ�Ĵ�ʩ��(������)
��4��װ��B�е���ˮ����Ϊ ��װ��C������Ϊ ��װ��D��FeCl2ȫ����Ӧ�����Ϊʧȥ����Cl2�����ö�ʧЧ��������ѡ������FeCl2�Ƿ���ȫʧЧ���Լ���___
| A������������Һ | B�����Ը��������Һ | C�����軯����Һ | D����ˮ |
��12�֣�ij��ȤС���о�SO2���廹ԭFe3+��I2������ʹ�õ�ҩƷ��װ������ͼ��ʾ��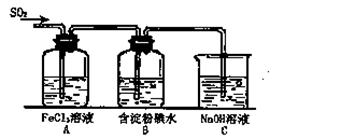
��1��SO2������Fe3+��Ӧ����Ҫ������__ __ ���������ӷ��ţ�
��2������ʵ�鷽������������ʵ������ȡ����SO2���� ��
| A��Na2SO3��Һ��HNO3 | B��Na2SO3������Ũ���� |
| C���������ڴ�����ȼ�� | D��ͭ����ŨH2SO4 |
��4�������280mL SO2���壨������Ϊ��̬������Cװ���У���C��50mL NaOH��Һ�����ʵ���Ũ������Ϊ mol/L���ܴﵽĿ�ġ�
��5��������װ����ͨ�������SO2��Ϊ����֤A��SO2��Fe3+������������ԭ��Ӧ������ȡA�е���Һ���ֳ����ݣ������������ʵ�飺
�����٣�����һ����Һ�м���KMnO4��Һ���Ϻ�ɫ��ȥ��
�����ڣ����ڶ�����Һ����KSCN��Һ������죬�ټ������Ƶ���ˮ����Һ��졣
�����ۣ�����������Һ������ϡ�����ữ��BaCl2��������ɫ������
������������������ ��ԭ���� ��
��6���ܱ���I���Ļ�ԭ������SO2�������� ��д���й����ӷ���ʽ�� ��
���������ռ�ʱӦʹ��������������
| A��ʯӢ���� | B����ͨ�������� | C�������� | D���մ����� |