��Ŀ����
11��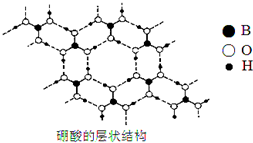 ��B�����仯�����ڻ�ѧ������Ҫ�ĵ�λ����ش��������⣺
��B�����仯�����ڻ�ѧ������Ҫ�ĵ�λ����ش��������⣺��1��Ga��Bͬ���壬Ga�Ļ�̬ԭ�Ӻ�������Ų�ʽΪ1s22s22p63s23p63d104s24p1��[Ar]3d104s24p1B��ͬ����������Ԫ�ص�һ�������ɴ�С��˳����C��Be��B��
��2�����ᣨH3BO3���ǰ�ɫƬ״���壨��״�ṹ��ͼ�����л���У�����ˮ���ܽ�Ⱥ�С������ʱ�ܽ������
��������Bԭ�ӵ��ӻ��������Ϊsp2��
�����ᾧ���д��ڵ��������з��»�������������ۼ���
�ۼ���ʱ��������ܽ��������Ҫԭ���Ǽ����ƻ����������֮�����������������ˮ����֮���γ������
��������һԪ���ᣬ��ˮ�е���ʱ��������ˮ�������OH���������ԣ�д������ĵ��뷽��ʽH3BO3+H2O?[B��OH��4]-+H+��
��3��������������һ�������մɲ��ϣ��ṹ�����嶼���ƽ��ʯ������ʱ��֪�ļ�����Ӳ�����ʣ���ѧʽΪBN����������������Bԭ�ӵ��ӻ��������Ϊsp3��1mol������������B-N�����ʵ���Ϊ4mol��
���� ��1��Ga��Bͬ���壬���ڵ������ڢ�A�壬���������Ϊ31����Ϻ�������Ų�������д��ͬ���ڴ����ҵ�һ�����������ǵ�IIA�͵�IIIA�塢��VA�͵�VIA������
��2������ͼ��֪��Bԭ���γ�3��B-O�Ҽ���û�йµ��Ӷԣ��ݴ��ж�Bԭ���ӻ���ʽ��
�������ᾧ��ṹ��֪�����ڷ�����O��B��H֮���γɹ��ۼ������Ӽ�H��O֮���γ������
�ۼ����ƻ����������֮�����������������ˮ����֮���γ������
��������һԪ���ᣬ��ˮ�е���ʱ��������ˮ�������OH-�������ԣ�����������[B��OH��4]-��H+��
��3������������Ľṹ�����嶼���ƽ��ʯ����ÿ��Bԭ���γ�4�����ۼ������ɵ�������״�ṹ�����ݹ��ۼ���Ŀ�ж��ӻ����ͣ�
��� �⣺��1��Ga��Bͬ���壬���ڵ������ڢ�A�壬���������Ϊ5+8+18=31�����ݹ���ԭ��֪���̬ԭ�Ӻ�����ӷֲ�ʽΪ1s22s22p63s23p63d104s24p1��[Ar]3d104s24p1��ͬ���ڴ����ҵ�һ�����������ǵ�IIA�͵�IIIA�塢��VA�͵�VIA�������ʵ�һ�����ܣ�C��Be��B��
�ʴ�Ϊ��1s22s22p63s23p63d104s24p1��[Ar]3d104s24p1�� C��Be��B��
��2������ͼ��֪��Bԭ���γ�3��B-O�Ҽ���û�йµ��Ӷԣ�Bԭ���ӻ������ĿΪ3��Bԭ�Ӳ�ȡsp2�ӻ���ʽ��
�ʴ�Ϊ��sp2��
�������ᾧ��ṹ��֪�����ڷ�����O��B��H֮���γɹ��ۼ������Ӽ�H��O֮���γ�����������֮��Ϊ���»�����
�ʴ�Ϊ����������ۼ���
�ۼ����ƻ����������֮�����������������ˮ����֮���γ����������ʱ��������ܽ������
�ʴ�Ϊ�������ƻ����������֮�����������������ˮ����֮���γ������
��������һԪ���ᣬ��ˮ�е���ʱ��������ˮ�������OH-�������ԣ�����������[B��OH��4]-��H+�����뷽��ʽΪ��H3BO3+H2O?[B��OH��4]-+H+��
�ʴ�Ϊ��H3BO3+H2O?[B��OH��4]-+H+��
��3������������Ľṹ�����嶼���ƽ��ʯ�����ʯ��ÿ��Cԭ��������4��̼ԭ���γ�������ṹ����������Ϊ������״�ṹ����ÿ��Bԭ���γ�4�����ۼ�����Bԭ�ӵ��ӻ�����Ϊsp3��ÿ��Bԭ���γ�4�����ۼ�����1mol������������B-N�����ʵ���Ϊ4mol��
�ʴ�Ϊ��sp3��4mol��
���� �����Ƕ����ʽṹ�Ŀ��飬��Ŀ�漰�����Ų�ʽ����һ�����ܡ��ӻ����۵�Ӧ������ͷ��Ӽ��������ȣ��漰֪ʶ��϶࣬��Ҫѧ���߱���ʵ�Ļ�������ʵǨ��������������Ŀ�Ѷ��еȣ�

 ֱͨ������У�ܲ��¿�ֱͨ��Уϵ�д�
ֱͨ������У�ܲ��¿�ֱͨ��Уϵ�д�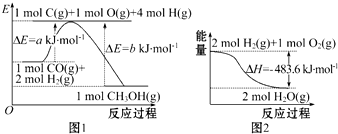
| A�� | ����ͼ1��֪�ϳɼ״����Ȼ�ѧ����ʽΪCO��g��+2H2��g���TCH3OH��g����H1=��b-a��kJ•mol-1 | |
| B�� | 1 mol NaOH�ֱ��1 mol CH3COOH��1 mol HNO3��Ӧ�����߱�ǰ�ߡ�HС | |
| C�� | ͼ2��ʾ2 mol H2��g�������е�������2 mol��̬ˮ�����е�������483.6 kJ | |
| D�� | ����ȼ��ʱ��ȫ���Ļ�ѧ��ת��Ϊ���� |
| A�� | �������Ȼ�̼����ˮ���ɼ��� | |
| B�� | CH3CH2OH��CH3COOH���������Na��Ӧ����H2 | |
| C�� | ���ࡢ�����ʡ���֬���ܷ���ˮ�ⷴӦ | |
| D�� | �����ʵ�������ϩ���Ҵ��ֱ���ȫȼ�գ�����O2��������� |
| A�� | ��������60 | B�� | ��������27 | C�� | ��������33 | D�� | ��������33 |
| A�� | Zn+CuSO4�TZnSO4+Cu | B�� | 2HI+Cl2�TI2+2HCl | ||
| C�� | CH4+Cl2$\stackrel{����}{��}$CH3Cl+HCl | D�� | CH4+2O2$\stackrel{��ȼ}{��}$CO2+2H2O |
�����Ͷȣ�����=˫����+��������2+���������磺
 ��=2������������
��=2�����й������л���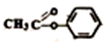 ��˵����ȷ���ǣ�������
��˵����ȷ���ǣ�������| A�� | �����Ͷ�Ϊ5 | |
| B�� | ���еĹ�������̼̼˫�������� | |
| C�� | ���л�����Է���ˮ�ⷴӦ�����ܷ���������Ӧ | |
| D�� | ���л�������ԭ�Ӷ���ͬһƽ���� |
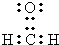 ��A������ԭ���Ƿ����ǣ���ǡ�����
��A������ԭ���Ƿ����ǣ���ǡ�����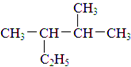 ��2��3-�������飻��
��2��3-�������飻��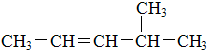 ��4-��-2-��ϩ��
��4-��-2-��ϩ��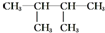 2��3-����-����
2��3-����-����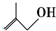 �ķ���ʽΪC4H8O��
�ķ���ʽΪC4H8O��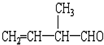 �����ŵ�������̼̼˫����ȩ����
�����ŵ�������̼̼˫����ȩ����