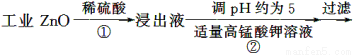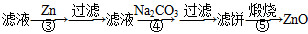ΧβΡΩΡΎ»ί
―–ΨΩΖΔœ÷Θ§Ω…“‘”Ο ·ΡΪΉς―τΦΪΓΔν―ΆχΉς“θΦΪΓΔ»έ»ΎCaF2ΓΣCaOΉςΒγΫβ÷ Θ§άϊ”Ο»γΆΦΥυ ΨΉΑ÷ΟΜώΒΟΫπ τΗΤΘ§≤Δ“‘ΗΤΈΣΜΙ‘≠ΦΝΜΙ‘≠Εΰ―θΜ·ν―÷Τ±ΗΫπ τν―ΓΘœ¬Ν–ΥΒΖ®÷–Θ§’ΐ»ΖΒΡ «Θ® Θ©

AΘ°”…TiO2÷ΤΒΟ1 molΫπ τTiΘ§άμ¬έ…œΆβΒγ¬ΖΉΣ“Τ2 molΒγΉ”
BΘ°―τΦΪΒΡΒγΦΪΖ¥”Π ΫΈΣCΘΪ2O2Θ≠Θ≠4eΘ≠=CO2Γϋ
CΘ°‘Ύ÷Τ±ΗΫπ τν―«ΑΚσΘ§’ϊΧΉΉΑ÷Ο÷–CaOΒΡΉήΝΩΦθ…Ό
DΘ°»τ”Ο«Π–νΒγ≥ΊΉςΗΟΉΑ÷ΟΒΡΙ©ΒγΒγ‘¥Θ§ΓΑΘΪΓ±Ϋ”œΏ÷υ”ΠΝ§Ϋ”PbΒγΦΪ
B
ΓΨΫβΈωΓΩΗυΨίΆΦ÷––≈œΔΘ§ΒγΦΪΖ¥”ΠΈΣ
―τΦΪΘΚCΘΪ2O2Θ≠Θ≠4eΘ≠=CO2Γϋ
“θΦΪΘΚ2Ca2ΘΪΘΪ4eΘ≠=2Ca
TiO2ΘΪ2Ca=TiΘΪ2CaO
AœνΘ§÷ΤΒΟ1 mol TiΘ§ΆβΒγ¬Ζ”ΠΉΣ“Τ4 mol eΘ≠ΘΜCœνΘ§÷Τ±Ην―«ΑΚσΘ§CaOΒΡΉήΝΩ±Θ≥÷≤Μ±δΘΜDœνΘ§ΓΑΘΪΓ±Ϋ”œΏ÷υ”ΠΫ”ΆβΫ”Βγ‘¥ΒΡ’ΐΦΪΘ§ Φ¥PbO2ΓΘ

 ΧλΧλœρ…œ“Μ±ΨΚΟΨμœΒΝ–¥πΑΗ
ΧλΧλœρ…œ“Μ±ΨΚΟΨμœΒΝ–¥πΑΗ –Γ―ß…ζ10Ζ÷÷””Π”ΟΧβœΒΝ–¥πΑΗ
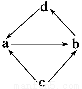
–Γ―ß…ζ10Ζ÷÷””Π”ΟΧβœΒΝ–¥πΑΗœ¬Ν–ΗςΉιΈο÷ ÷–Θ§Έο÷ ÷°ΦδΆ®Ιΐ“Μ≤ΫΖ¥”ΠΨΆΡή Βœ÷ΆΦ Ψ±δΜ·ΒΡ «(ΓΓΓΓ)
Έο÷ ±ύΚ≈ | Έο÷ ΉΣΜ·ΙΊœΒ | a | b | c | d |
ΔΌ |
| FeCl2 | FeCl3 | Fe | CuCl2 |
ΔΎ | NO | NO2 | N2 | HNO3 | |
Δέ | Na2O | Na2O2 | Na | NaOH | |
Δή | Al2O3 | NaAlO2 | Al | Al(OH)3 |
AΘ°ΔΌΔή BΘ°ΔΌΔΎΔέ CΘ°ΔΌΔέΔή DΘ°ΔΎΔή
”–“ΜΈό…ΪΆΗΟς»ή“ΚΘ§”ϊ»ΖΕ® «ΖώΚ§”–œ¬Ν–άκΉ”ΘΚKΘΪΓΔMg2ΘΪΓΔAl3ΘΪΓΔFe2ΘΪΓΔBa2ΘΪΓΔNO3-ΓΔSO42-ΓΔClΘ≠ΓΔIΘ≠ΓΔHCO3-Θ§»ΓΗΟ»ή“ΚΫχ––»γœ¬ Β―ιΘΚ
Β―ι≤Ϋ÷η | Β―ιœ÷œσ |
ΔΌ»Γ…ΌΝΩΗΟ»ή“ΚΘ§Φ”ΦΗΒΈΦΉΜυ≥»»ή“Κ | »ή“Κ±δΚλ…Ϊ |
ΔΎ»Γ…ΌΝΩΗΟ»ή“ΚΘ§Φ”»κΆ≠Τ§ΚΆ≈®ΝρΥαΘ§Φ”»» | ”–Έό…ΪΤχΧε≤ζ…ζΘ§”ωΩ’ΤχΩ…“‘±δ≥…ΚλΉΊ…Ϊ |
Δέ»Γ…ΌΝΩΗΟ»ή“ΚΘ§Φ”»κBaCl2»ή“Κ | ”–ΑΉ…Ϊ≥ΝΒμ…ζ≥… |
Δή»ΓΔέ÷–ΒΡ…œ≤ψ«ε“ΚΘ§Φ”»κAgNO3»ή“Κ | ”–Έ»Ε®ΒΡΑΉ…Ϊ≥ΝΒμ…ζ≥…Θ§«“≤Μ»ή”ΎœΓœθΥα |
Δί»Γ…ΌΝΩΗΟ»ή“ΚΘ§Φ”»κNaOH»ή“Κ | ”–ΑΉ…Ϊ≥ΝΒμ…ζ≥…Θ§Β±NaOHΙΐΝΩ ±Θ§≥ΝΒμ≤ΩΖ÷»ήΫβ |
”…¥Υ≈–ΕœΘΚ
Θ®1Θ©»ή“Κ÷–“ΜΕ®¥φ‘ΎΒΡάκΉ” «______________ΘΜ»ή“Κ÷–ΩœΕ®≤Μ¥φ‘ΎΒΡάκΉ” «________________________ΓΘ
Θ®2Θ©ΈΣΫχ“Μ≤Ϋ»ΖΕ®ΤδΥϊάκΉ”Θ§”ΠΗΟ≤Ι≥δΒΡ Β―ιΦΑΕ‘”Π”ϊΦλ―ιάκΉ”ΒΡΟϊ≥Τ(»τΈΣ»ή“ΚΖ¥”ΠΘ§ΥΒΟς Ι”Ο ‘ΦΝΒΡΟϊ≥ΤΘ§≤Μ±Ί–¥œξœΗ≤Ϋ÷η)________________________________________ΓΘ
Θ®3Θ©–¥≥ω Β―ιΔί÷–Υυ”–Ζ¥”ΠΒΡάκΉ”ΖΫ≥Χ ΫΘΚ______________________________ΓΘ