��Ŀ����
����Ŀ����֪����pHΪ4��5�Ļ����У�Cu2����Fe2��������ˮ�⣬��Fe3��������ȫˮ�⡣�ô�����ͭ��CuO�к�����Fe����ȡCuCl2��Һ�������£�
��ȡһ�����Ĵ�CuO���������Ũ���ᣬ���Ȳ����裬��ַ�Ӧ����ˣ���Һ��pHΪ1��
������Һ��ͨ��Cl2�����衣
�������м�����������A���ȣ����裬������Һ��pH�����ˡ�
������������ҺŨ����
��1�����м��Ȳ������Ŀ����_____________________________��
��2�����з�����Ӧ�����ӷ���ʽ��_________________________��
��3�����е�����A������__________________��������PH��Χ��_______________��
���˺������ijɷ���__________________��
��4�����з�����Ӧ�����ӷ���ʽ��_________________________��
��5���٢��������pH��ֽ�ⶨ��Һ��pHֵ��_________________________��
���𰸡��ӿ췴Ӧ���ʣ��Ƿ�Ӧ��� 2Fe2++Cl2=2Fe3++2Cl- CuO 4-5 Fe(OH)3 CuO+2H+=Cu2++H2O ȡһС��pH��ֽ�����ڲ���Ƭ�ϣ��ò�����պȡ����Һ������pH��ֽ���룬����ɫ�������ɫ����ȡ�
��������
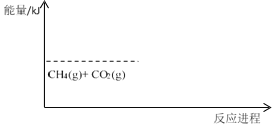
����ͭ��Ʒ����Ũ�����ܽ⣬��ʱ��Һ����Ҫ��Cu2+��Fe2+��Cl-��H+��ͨ����������������������Fe3+�����ӷ���ʽΪ��![]() ����������CuO���Ե���pHֵ��4-5����Fe3+ת���ɳ�����ͬʱ�������µ����ʣ�֮�����Ũ�����õ�CuCl2��Һ��
����������CuO���Ե���pHֵ��4-5����Fe3+ת���ɳ�����ͬʱ�������µ����ʣ�֮�����Ũ�����õ�CuCl2��Һ��
��1�����Ƚ���һ����Ϊ�˼ӿ췴Ӧ�ٶȣ�
�ʴ�Ϊ���ӿ췴Ӧ���ʣ�ʹ��Ӧ��֣�
��2�����ݷ�����֪��Ϊ��![]() ��
��
��3�����ݷ�����֪AΪCuO�����ܵ���pHֵ�ֲ������µ����ʣ�������Ŀ��Ϣ��֪pH��4-5�Ļ�����Cu2��������ˮ�⣬��Fe3��������ȫˮ�⣻�õ�������ΪFe(OH)3��
�ʴ�Ϊ��CuO��4-5��Fe(OH)3��
��4���÷�ӦΪ����ͭ�������ӵķ�Ӧ��
�ʴ�Ϊ��CuO+2H+=Cu2++H2O��
��5������ҺpHֵ�ķ���Ϊ��ȡһС��pH��ֽ�����ڲ���Ƭ�ϣ��ò�����պȡ����Һ������pH��ֽ���룬����ɫ�������ɫ����ȡ�