��Ŀ����
19���±���Ԫ�����ڱ���һ���֣���Ա��еĢ١�����Ԫ�أ���ջش�| ���� ���� | ��A | ��A | ��A | ��A | ��A | ��A | ��A | 0 |
| �� | �� | �� | �� | |||||
| �� | �� | �� | �� | �� | �� |
 ��
����2���ۢݢ�Ԫ��ԭ���γɼ����ӵ����Ӱ뾶�ɴ�С��˳����Cl-��F-��Al3+�������ӷ��ţ���
��3��ijԪ�ض��������ӵĺ�����10�����ӣ���Ԫ����þ����Ԫ�����ƣ���
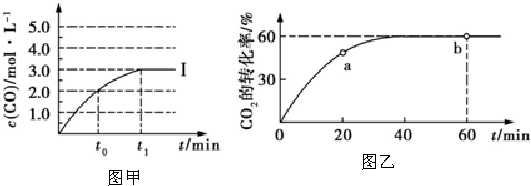
��4���ܵĵ�����ˮ��Ӧ�Ļ�ѧ����ʽ��
 �����������ת�Ƶķ�������Ŀ����
�����������ת�Ƶķ�������Ŀ������5����Ԫ�صĵ��ʳ����µ���ɫ�ǻ���ɫ���õ���ʽ��ʾ��Ԫ�ص��⻯���γɹ���Ϊ
 ��
����6����֪ijЩ��ͬ��Ԫ�ص�����Ҳ��һ���������ԣ���Ԫ�آ����Ԫ�ص��������������Ƶ����ʣ�д��Ԫ�آٵ�����������NaOH��Һ��Ӧ�Ļ�ѧ����ʽBe��OH��2+2NaOH=Na2BeO2+2H2O��
���� ��Ԫ�������ڱ��е�λ�ÿ�֪����ΪBe����ΪN����ΪF����ΪNa����ΪAl����ΪSi����ΪCl����ΪAr��
��1��ϡ������Ar�Ļ�ѧ��������ã�
��2�����Ӳ�ṹ��ͬ�����ӣ��˵����Խ�����Ӱ뾶ԽС�����ӵ��Ӳ�Խ�����Ӱ뾶Խ��
��3��ijԪ�ض��������ӵĺ�����10�����ӣ���Ԫ��ԭ����12�����ӣ���Ԫ��ΪMg��
��4��Na��ˮ��Ӧ����������������������Ӧת�Ƶ�����Ϊ2e-��
��5������Ϊ����ɫ���壬HCl���ڹ��ۻ��������ԭ�ӡ���ԭ�ӵ���ʽ��ʾ���γɹ��̣�
��6��Be��OH��2�������������������ƣ���Be��OH��2���������Ʒ�Ӧ����Na2BeO2��ˮ��
��� �⣺��Ԫ�������ڱ��е�λ�ÿ�֪����ΪBe����ΪN����ΪF����ΪNa����ΪAl����ΪSi����ΪCl����ΪAr��
��1��ϡ������Ar�Ļ�ѧ��������ã���ԭ�ӽṹʾ��ͼΪ ��
��
�ʴ�Ϊ�� ��
��
��2�����Ӳ�ṹ��ͬ�����ӣ��˵����Խ�����Ӱ뾶ԽС�����ӵ��Ӳ�Խ�����Ӱ뾶Խ�����Ӱ뾶�ɣ�Cl-��F-��Al3+��
�ʴ�Ϊ��Cl-��F-��Al3+��
��3��ijԪ�ض��������ӵĺ�����10�����ӣ���Ԫ��ԭ����12�����ӣ���Ԫ��ΪMg��Ԫ������Ϊþ��
�ʴ�Ϊ��þ��
��4��Na��ˮ��Ӧ����������������������Ӧת�Ƶ�����Ϊ2e-���������ת����Ŀ�뷽��Ϊ�� ��
��
�ʴ�Ϊ�� ��
��
��5������Ϊ����ɫ���壬HCl���ڹ��ۻ��������ԭ�ӡ���ԭ�ӵ���ʽ��ʾ���γɹ���Ϊ��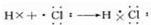 ��
��
�ʴ�Ϊ������ɫ��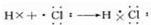 ��
��
��6��Be��OH��2�������������������ƣ���Be��OH��2���������Ʒ�Ӧ����Na2BeO2��ˮ����Ӧ����ʽΪ��Be��OH��2+2NaOH=Na2BeO2+2H2O��
�ʴ�Ϊ��Be��OH��2+2NaOH=Na2BeO2+2H2O��
���� ������Ԫ�����ڱ���Ԫ�������ɣ��ѶȲ���ע���Ԫ�������ɵ��������գ�ע���õ���ʽ��ʾ���ʻ�ѧ�����γɣ�

 �»����ܶ�Ա��ϵ�д�
�»����ܶ�Ա��ϵ�д� ����ͼ����ּ��������ҵ֣�ݴ�ѧ������ϵ�д�
����ͼ����ּ��������ҵ֣�ݴ�ѧ������ϵ�д��� ���� | ��A | ��A | ��A | ��A | ��A | ��A | ��A | 0 |
| �� | �� | �� | ||||||
| �� | �� | �� | �� | �� | �� | �� | ||
| �� | �� | �� |
 ��
����2���õ���������ǿ����F �ؿ��к������Ľ���Ԫ����Al��
��3���õ���ʽ��ʾ������γɻ�����Ĺ���
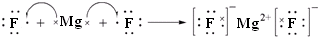 ��
����4����ЩԪ���е�����������Ӧ��ˮ�����У�������ǿ����HClO4��������ǿ����KOH�������Ե�����������Al��OH��3��
��5���ȽϢڢۢܢ��γɵļ����Ӱ뾶�Ĵ�СCl-��F-��Na+��Mg2+��
��6��ijԪ��R����̬�⻯��ΪHxR����R�ڸ��⻯���е���������Ϊ94%��8.5g��HxR�����ڱ�״̬�µ������5.6L����HxR����Է�����Ϊ34��HxR�Ļ�ѧʽΪH2S��
| �� ���� | ��A | ��B | ��A | ��A | ��A | ��A | ��A | ||
| һ | �� | ||||||||
| �� | �� | �� | �� | �� | �� | ||||
| �� | �� | �� | �� | �� | |||||
| �� | ⑪ | ⑫ | ⑬ | ||||||
��2������Ԫ�آ��ԭ�ӽṹʾ��ͼ��
 ��
����3������Ԫ�آޢ���⻯����ȶ���˳��ΪHF��HCl��H2S����д��ѧʽ����ͬ����
��4������Ԫ�آ�͢�����������Ӧˮ��������ԣ�HClO4��H2SO4��
 ʵ����������0.2000mol•L -1Na2S2O3����Һ450mL�������ø���Һ��ijŨ�ȵ�NaClO��Һ���б궨��
ʵ����������0.2000mol•L -1Na2S2O3����Һ450mL�������ø���Һ��ijŨ�ȵ�NaClO��Һ���б궨����1������Na2S2O3���������Ʊ���Һ������ͼ��ʾ�������У�����Ҫ�õ���������AB������ĸ������ȱ�ٵIJ����������ձ��������������������ƣ���
��2�����ݼ���������ƽ��ȡNa2S2O3�����������15.8g����ʵ����������������ȷ��������ƿ������ˮϴ�Ӻ�δ�����������ҺŨ��=���������������=������ͬ��0.2000mol•L-1������δ����Һ��ȴ�Ͷ����ˣ���������ҺŨ�ȣ�0.2000mol•L-1��
��3���õζ����궨�ľ��巽������ȡ20.00mL NaClO��Һ����ƿ�У���������ϡ���������KI���壬��0.2000mol•L -1 Na2S2O3����Һ�ζ����յ㣨������Һ��ָʾ�������Ĵ�ƽ��ʵ��ⶨ��V��Na2S2O3���������£�
����֪��I2+2Na2S2O3�T2NaI+Na2S4O6��
| �ⶨ���� | ��һ�� | �ڶ��� | ������ | ���Ĵ� |
| V��Na2S2O3��/mL | 21.90 | 18.80 | 22.10 | 22.00 |
��NaClO��Һ�����ʵ���Ũ����0.1060 mol•L -1��

| A�� | װ��I��װ��II�и�����Ӧ���ǣ�Fe-2e-=Fe2+ | |
| B�� | װ��I��װ��II�������е������Ӿ����Ҳ��ձ��ƶ� | |
| C�� | װ��I��װ��II��Fe�缫 �ĸ�ʴ�ٶ�Ϊǰ�ߴ��ں��� | |
| D�� | �ŵ�����У�װ��I����ձ���װ��II�Ҳ��ձ�����Һ��PH������ |