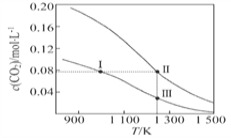
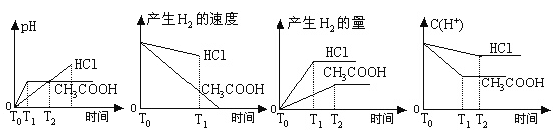
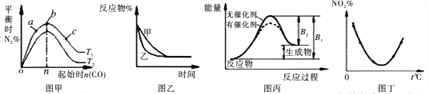
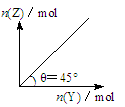ЬтФПФкШн
ЁОЬтФПЁПвбжЊЫЎдк25ЁцКЭTЁцЪБЃЌЦфЕчРыЦНКтЧњЯпШчЭМЫљЪОЃК
ЃЈ1ЃЉ25ЁцЪБЃЌгаpHЃН12ЕФBa(OH)2ШмвКЁЃ
ЂйИУBa(OH)2ШмвКЕФЮяжЪЕФСПХЈЖШЮЊ__________________ЃЛ
Ђк25ЁцЪБЃЌЯђ100mLpH=2ЕФСђЫсгыСђЫсФЦЕФЛьКЯШмвКжаЃЌМгШы300mLЩЯЪіBa(OH)2ШмвКЃЌГСЕэе§КУДязюДѓСПЃЈ МйЩшШмвКЛьКЯЧАКѓЕФЬхЛ§БфЛЏКіТдВЛМЦЃЉЃЌдЛьКЯШмвКжаСђЫсФЦЕФЮяжЪЕФСПХЈЖШЮЊ_______________ЁЃ
ЃЈ2ЃЉTЁцЪБЃЌНЋ100 mL pHЃН5ЕФСђЫсШмвКЯЁЪЭжСдЬхЛ§ЕФ1000БЖЃЌЯЁЪЭКѓШмвКжаcЃЈH+ЃЉ/cЃЈSO42-ЃЉЃН____________ЁЃ
ЃЈ3ЃЉTЁцЪБЃЌНЋ0ЃЎ1 molЁЄLЃ1ЕФNaHSO4ШмвКгы0.1 molЁЄLЃ1ЕФBaЃЈOHЃЉ2ШмвКАДЯТБэжаМзЁЂввЁЂБћЁЂЖЁВЛЭЌЗНЪНЛьКЯЃК
Мз | вв | Бћ | ЖЁ | |
0ЃЎ1 molЁЄLЃ1 BaЃЈOHЃЉ2ШмвКЬхЛ§/mL | 10 | 10 | 10 | 10 |
0ЃЎ1 molЁЄLЃ1 NaHSO4ШмвКЬхЛ§/mL | 5 | 10 | 15 | 20 |
ЂйАДЖЁЗНЪНЛьКЯКѓЃЌЫљЕУШмвКЯд________________ЃЈЬюЁАЫсЁБЁЂЁАМюЁБЛђЁАжаЁБЃЉадЁЃ
ЂкаДГіАДввЗНЪНЛьКЯКѓЃЌЗДгІЕФРызгЗНГЬЪНЃК_________________________ЁЃ
ЂлАДМзЗНЪНЛьКЯКѓЃЌЫљЕУШмвКЕФpHЮЊ______________________ЁЃ
ЁОД№АИЁП 5ЁС10-3molЁЄL-1 0.01molЁЄL-1 200ЁУ1ЃЈЛђ200ЃЉ жа Ba2++OH-+H++SO42-=BaSO4Ё§+H2O 11
ЁОНтЮіЁПЃЈ1ЃЉЂй25ЁцЪБЃЌгаpHЃН12ЕФBa(OH)2ШмвКжаc(OH-)=10-2mol/LЃЌc[Ba(OH)2]=![]() c(OH-)=
c(OH-)=![]() ЁС10-2mol/L=5ЁС10-3molЁЄL-1ЃЛЂк300mL5ЁС10-3molЁЄL-1Ba(OH)2ШмвКЕФЮяжЪЕФСПЮЊЃК0.30LЁС5ЁС10-3molЁЄL-1=1.5ЁС10-3molЃЌ100mLpH=2ЕФСђЫсжаn(SO42-)=
ЁС10-2mol/L=5ЁС10-3molЁЄL-1ЃЛЂк300mL5ЁС10-3molЁЄL-1Ba(OH)2ШмвКЕФЮяжЪЕФСПЮЊЃК0.30LЁС5ЁС10-3molЁЄL-1=1.5ЁС10-3molЃЌ100mLpH=2ЕФСђЫсжаn(SO42-)=![]() =0.5ЁС10-3mol ЃЌдђСђЫсФЦЕФЮяжЪЕФСПЮЊЃК1.5ЁС10-3mol-0.5ЁС10-3mol=1.0ЁС10-3molЃЌc(Na2SO4)=
=0.5ЁС10-3mol ЃЌдђСђЫсФЦЕФЮяжЪЕФСПЮЊЃК1.5ЁС10-3mol-0.5ЁС10-3mol=1.0ЁС10-3molЃЌc(Na2SO4)=![]() =0.01molЁЄL-1ЃЛЃЈ2ЃЉЪвЮТЪБНЋPH=5ЕФСђЫсШмвКЯЁЪЭ1000БЖКѓc(H+)Ёж1.0ЁС10-7mol/LЃЛc(SO42-)=
=0.01molЁЄL-1ЃЛЃЈ2ЃЉЪвЮТЪБНЋPH=5ЕФСђЫсШмвКЯЁЪЭ1000БЖКѓc(H+)Ёж1.0ЁС10-7mol/LЃЛc(SO42-)=![]() ЁС10-8mol/LЃЛЫљвдЕУc(H+):c(SO42-)=20:1ЃЛЃЈ3ЃЉЂйАДЖЁЗНЪНЛьКЯЪБЃЌBa(OH)2ЬсЙЉЕФOH-гыNaHSO4ЬсЙЉЕФH+ЯрЕШЃЌЛьКЯКѓШмвКГЪжаадЃЛЂкАДввЗНЪНЛьКЯЪБЃЌЗДгІЧАЃКnЃЈBa2+ЃЉ=10-3molЃЌnЃЈOH-ЃЉ=2ЁС10-3molЃЌnЃЈH+ЃЉ=nЃЈSO42-ЃЉ=10-3molЃЌЪЕМЪЗДгІЕФBa2+ЁЂOH-ЁЂH+ЁЂSO42- ОљЮЊ10-3molЃЌЙЪЗДгІЕФРызгЗНГЬЪНЮЊBa2++OH-+H++SO42-=BaSO4Ё§+H2OЃЛЂлАДМзЗНЪНЛьКЯЪБЃЌOH-Й§СПЃЌЗДгІКѓШмвКжаcЃЈOH-ЃЉ=
ЁС10-8mol/LЃЛЫљвдЕУc(H+):c(SO42-)=20:1ЃЛЃЈ3ЃЉЂйАДЖЁЗНЪНЛьКЯЪБЃЌBa(OH)2ЬсЙЉЕФOH-гыNaHSO4ЬсЙЉЕФH+ЯрЕШЃЌЛьКЯКѓШмвКГЪжаадЃЛЂкАДввЗНЪНЛьКЯЪБЃЌЗДгІЧАЃКnЃЈBa2+ЃЉ=10-3molЃЌnЃЈOH-ЃЉ=2ЁС10-3molЃЌnЃЈH+ЃЉ=nЃЈSO42-ЃЉ=10-3molЃЌЪЕМЪЗДгІЕФBa2+ЁЂOH-ЁЂH+ЁЂSO42- ОљЮЊ10-3molЃЌЙЪЗДгІЕФРызгЗНГЬЪНЮЊBa2++OH-+H++SO42-=BaSO4Ё§+H2OЃЛЂлАДМзЗНЪНЛьКЯЪБЃЌOH-Й§СПЃЌЗДгІКѓШмвКжаcЃЈOH-ЃЉ=![]() =0.1mol/LЃЌcЃЈH+ЃЉ=10-11mol/LЃЌШмвКЕФpH=11ЁЃ
=0.1mol/LЃЌcЃЈH+ЃЉ=10-11mol/LЃЌШмвКЕФpH=11ЁЃ