��Ŀ����
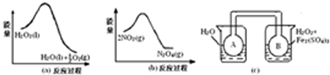
14��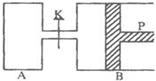 ͼ�У�PΪһ�����ɻ����Ļ������ر�K���ֱ�������A��B�и�����2molX��2molY����ʼʱ��VA=aL��VB=0.8aL����ͨ�ܵ�������Բ��ƣ�������ͬ�¶Ⱥ��д������ڵ������£��������и��Է���������Ӧ��
ͼ�У�PΪһ�����ɻ����Ļ������ر�K���ֱ�������A��B�и�����2molX��2molY����ʼʱ��VA=aL��VB=0.8aL����ͨ�ܵ�������Բ��ƣ�������ͬ�¶Ⱥ��д������ڵ������£��������и��Է���������Ӧ��3X��g��+3Y��g��?2Z��g��+2W��g�����ﵽƽ��ʱ��VB=0.6aL��
��1��ƽ��ʱA��B�л�������ƽ����Է��������Ĺ�ϵ�ǣ�MA��MB���������=������������
��2�����Ҫ�����ƽ��ʱB�л��������ܶȣ������ٻ���Ҫ֪����������A��B��MX��MY��MZ��MW
�ֱ��ʾX��Y��Z��W��Ħ������������MX ��MY ��MZ ��MW
A���ۺ͢�B���ٺ͢�C���ٺ͢�D���ڢۢ�
��3����K��һ��ʱ���Ӧ�ٴδﵽƽ�⣬��B�����Ϊ0.2aL��
���� ��1����ʼʱA����������A��ѹǿС������ӦΪ�������ʵ�����С�ķ�Ӧ��AΪ���º��ݣ�ƽ���ѹǿ��С��BΪ���º�ѹ����ЧΪ��Aƽ��Ļ���������ѹǿ����A��ƽ����ȣ�ƽ�������ƶ�����ƽ��ʱB��������������ʵ�����A�е�С��������������������䣬�ٸ���M=$\frac{m}{n}$�жϣ�
��2����������ܶ�=������������������ݻ��������ݻ���֪����Ҫ����������������������������������ʼʱX��Y����֮�ͣ�Ҳ���Ե�ЧΪ��ʼ����$\frac{4}{3}$molZ��$\frac{4}{3}$molW�����ƽ�⣬�������������������ʼʱZ��W����֮�ͣ�
��3����K��һ��ʱ���Ӧ�ٴδﵽƽ�⣬��ЧΪ��ʼ����4molX��4molY�����ƽ�⣬���º�ѹ����B��ƽ��Ϊ��Чƽ�⣬��Ӧ��ת���ʲ��䣬ƽ��ʱ������������ʵ���ΪB��2�������º�ѹ��ѹǿ֮�ȵ������ʵ���֮�ȣ��������㿪ʼ����4molX��4molY�����ƽ��ʱ���������ȥ����A�������ΪB���������
��� �⣺��1����ʼʱA����������A��ѹǿС������ӦΪ�������ʵ�����С�ķ�Ӧ��AΪ���º��ݣ�ƽ���ѹǿ��С��BΪ���º�ѹ����ЧΪ��Aƽ��Ļ���������ѹǿ����A��ƽ����ȣ�ƽ�������ƶ�����ƽ��ʱB��������������ʵ�����A�е�С��������������������䣬�ٸ���M=$\frac{m}{n}$��֪��MA��MB��
�ʴ�Ϊ������
��2����������ܶ�=������������������ݻ��������ݻ���֪����Ҫ����������������������������������ʼʱX��Y����֮�ͣ�Ҳ���Ե�ЧΪ��ʼ����$\frac{4}{3}$molZ��$\frac{4}{3}$molW�����ƽ�⣬�������������������ʼʱZ��W����֮�ͣ�����Ҫ���ٻ���Ҫ֪���������ǣ�X��YĦ��������Z��WĦ��������
��ѡ��A��B��
��3����K��һ��ʱ���Ӧ�ٴδﵽƽ�⣬��ЧΪ��ʼ����4molX��4molY�����ƽ�⣬���º�ѹ����B��ƽ��Ϊ��Чƽ�⣬��Ӧ��ת���ʲ��䣬ƽ��ʱ������������ʵ���ΪB��2�������º�ѹ��ѹǿ֮�ȵ������ʵ���֮�ȣ���ʼ����4molX��4molY�����ƽ��ʱ�����Ϊ0.6aL��2=1.2aL����B�������Ϊ1.2aL-aL=0.2aL��
�ʴ�Ϊ��0.2a��
���� ���⿼�黯ѧƽ����㣬�漰��Чƽ�����⣬�ϺõĿ���ѧ��������������������ؼ��ǵ�Чƽ��;���Ľ������Ѷ��еȣ�

| A�� | MgSO4 ��Һ��Ba��OH��2��Һ��Ӧ��SO42-+Ba2+�TBa SO4�� | |
| B�� | ����ˮ�ķ�Ӧ��2Na+2H2O�T2Na++2OH-+H2�� | |
| C�� | ��Ƭ������������Һ��Ӧ��2Al+2OH--+2H2O�TAlO2-+3H2�� | |
| D�� | �������������������Fe��OH��3+3H+�TFe3++3H2O |
��1���ڱ�״���£���a L��SO2��Cl2��ɵĻ������ͨ��200mL 0.1mol/L��Fe2��SO4��3��Һ�У���ַ�Ӧ����Һ���ػ�ɫ��dz����Ӧ�����Һ�м���������BaCl2��Һ�������ó������ˡ�ϴ�ӡ��������أ�������Ϊ23.3g������������SO2�����Ϊ0.896L��a��ȡֵ��ΧΪ��1.344��a��1.792��
��2��ij�¶��£�SO2��g��+$\frac{1}{2}$O2��g��?SO3��g����H=-98kJ•mol-1����ʼʱ��100L���ܱ������м���4.0
mol SO2��g����10.0mol O2��g��������Ӧ�ﵽƽ��ʱ���ų�����196kJ�����¶���ƽ�ⳣ��K=3.33��
��3��һ�������£���һ���������ܱ������г���2mol SO2��1mol O2���������з�Ӧ��
2SO2��g��+O2��g��?2SO3��g�����ﵽƽ���ı�����������SO2��O2��SO3����ƽ��Ũ�ȶ���ԭ���������ACF������ĸ����
A�������¶Ⱥ�����������䣬����2mol SO3
B�������¶Ⱥ�����������䣬����2mol N2
C�������¶Ⱥ�����������䣬����0.5mol SO2��0.25mol O2
D�������¶Ⱥ�������ѹǿ���䣬����1mol SO3
E�������¶�
F���ƶ�����ѹ������
��4������ʱ��BaSO4��Ksp=1.08��10-10���ֽ��������BaCl2��Һ��2.0��10-3mol/L��Na2SO4��Һ��ϣ���Ҫ����BaSO4������BaCl2��Һ����СŨ��Ϊ2.16��10-7mol/L��
��5��N2O5��һ�������������������ʺ��Ʊ��ܵ����ǵĹ�ע����һ���¶��£��ں����ܱ�������N2O5�ɷ������з�Ӧ��2N2O5��g��?4NO2��g��+O2��g����H��0��
���Ϊ��Ӧ��T1�¶��µIJ���ʵ�����ݣ�
| t/s | 0 | 50 | 100 |
| c��N2O5��/mol•L-1 | 5.0 | 3.5 | 2.4 |
������H2��O2��������Na2CO3��ɵ�ȼ�ϵ�أ����õ�ⷨ�Ʊ�N2O5��װ����ͼ��ʾ������YΪCO2��
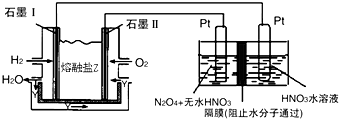
д��ʯīI�缫�Ϸ�����Ӧ�ĵ缫��ӦʽH2+CO32--2e-=CO2+H2O��
 ������Ԫ��X��Y��Z��������Ϊ1��7��12�γɵĻ����������������ӵ����ӷ�Ӧ����ʽ�ǣ�
������Ԫ��X��Y��Z��������Ϊ1��7��12�γɵĻ����������������ӵ����ӷ�Ӧ����ʽ�ǣ�