��Ŀ����
5��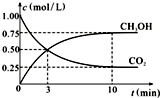 �״�����Ҫ�Ļ���ԭ�ϣ��ֿɳ�Ϊȼ�ϣ����úϳ�������Ҫ�ɷ�ΪCO��CO2��H2���ڴ����������ºϳɼ״�������������Ӧ���£�
�״�����Ҫ�Ļ���ԭ�ϣ��ֿɳ�Ϊȼ�ϣ����úϳ�������Ҫ�ɷ�ΪCO��CO2��H2���ڴ����������ºϳɼ״�������������Ӧ���£���CO��g��+2H2��g��?CH3OH��g����H1
��CO2��g��+3H2��g��?CH3OH��g��+H2O��g����H2
��CO2��g��+H2��g��?CO��g��+H2O��g����H3
�ش��������⣺
��1����֪��Ӧ���е���صĻ�ѧ�������������£�������CO�е�̼����ΪC$\frac{\underline{\;��\;}}{\;}$O��
| ��ѧ�� | H-H | C-O | C$\frac{\underline{\;��\;}}{\;}$O | H-O | C-H |
| E/��kJ��mol-1�� | 436 | 343 | 1076 | 465 | 413 |
��2����25�桢101KPa�£�ÿ���ȼ��1g CH3OH���ָ���ԭ״̬�����ͷ�22.68KJ����������д����ʾ�״�ȼ���ȵ��Ȼ�ѧ��Ӧ����ʽ��CH3OH��l��+$\frac{3}{2}$O2��g��=CO2��g��+2H2O��l����H=-725.76kJ•mol-1��
��3�����ڵڶ�����Ӧ����ʽ��CO2��g��+3H2��g��?CH3OH��g��+H2O��g������һ���º����ܱ������г���1molCO2��3mol H2������������Ӧ�����CO2��CH3OH��g����Ũ����ʱ��仯��ͼ��ʾ���Իش�0��10min�ڣ�������ƽ����Ӧ����Ϊ0.225mol•L-1•mm-1���÷�Ӧƽ�ⳣ������ʽ$\frac{c��C{H}_{3}OH��•c��{H}_{2}O��}{c��C{O}_{2}��•{c}^{3}��{H}_{2}��}$����10min��������������ٳ���1mol CO2��3mol H2�����ٴδﵽƽ��ʱCH3OH��g�����������������������١����䡱�����÷�Ӧ��ƽ�ⳣ�����䣨��������С�����䡱����
���� ��1����Ӧ��=��Ӧ���ܼ���-�������ܼ��ܣ����ݸ�˹���ɣ���Ӧ��-��Ӧ��=��Ӧ�ۣ���Ӧ��Ҳ������Ӧ�ļ��㣻
��2������ȼ���ȸ�����1mol��ȼ����ȫȼ�������ȶ�������ų����������������������32g�״�ȼ�����ɶ�����̼��Һ̬ˮ���ȣ�����Ȼ�ѧ����ʽ��д��������ע���ʾۼ�״̬�Ͷ�Ӧ�ʱ䣻
��3������v=$\frac{��c}{��t}$������ƽ��ʱ������Ũ����֮�����Ϸ�Ӧ��Ũ����֮������10min��������������ٳ���1mol CO2��3mol H2���൱������ѹǿ��ƽ�������ƶ���ƽ�ⳣ�����¶ȵĺ������ɴ˷������
��� �⣺��1����Ӧ��=��Ӧ���ܼ���-�������ܼ��ܣ��ʡ�H1=1076kJ��mol-1+2��436kJ��mol-1-��3��413+343+465��kJ��mol-1=-99kJ��mol-1��
���ݸ�˹���ɣ���Ӧ��-��Ӧ��=��Ӧ�ۣ��ʡ�H3=��H2-��H1=-58kJ��mol-1-��-99kJ��mol-1��=+41kJ��mol-1��
�ʴ�Ϊ��-99��+41��
��2����25�桢101kPa�£�1g�״���CH3OH��ȼ������CO2��Һ̬ˮʱ����22.68kJ��32g�״�ȼ�����ɶ�����̼��Һ̬ˮ�ų�����Ϊ725.76KJ�����ʾ�״�ȼ���ȵ��Ȼ�ѧ����ʽΪ��CH3OH��l��+$\frac{3}{2}$O2��g��=CO2��g��+2H2O��l����H=-725.76kJ•mol-1��
�ʴ�Ϊ��CH3OH��l��+$\frac{3}{2}$O2��g��=CO2��g��+2H2O��l����H=-725.76kJ•mol-1
��3��v��CH3OH��=$\frac{0.75mol/L}{10min}$=0.075mol/��L•min������v��H2��=3v��CH3OH��=0.075mol/��L•min����3=0.225mol•L-1•mm-1��ƽ�ⳣ��K=$\frac{c��C{H}_{3}OH��•c��{H}_{2}O��}{c��C{O}_{2}��•{c}^{3}��{H}_{2}��}$���൱������ѹǿ��ƽ�������ƶ����ٴδﵽƽ��ʱCH3OH��g��������������ƽ�ⳣ�����¶ȵĺ������¶Ȳ��䣬ƽ�ⳣ�����䣬�ʴ�Ϊ��0.225mol•L-1•mm-1��$\frac{c��C{H}_{3}OH��•c��{H}_{2}O��}{c��C{O}_{2}��•{c}^{3}��{H}_{2}��}$������䣮
���� ���⿼�鷴Ӧ���йؼ��㡢ƽ�ⳣ������Ӱ�����ء���ѧƽ���Ӱ�����ء���ѧƽ��ͼ���ۺ�Ӧ�õȣ����ؿ���ѧ������������������Ҫѧ���߱���ʵ�Ļ������Ѷ��еȣ�

| A�� | ÿ����1mol O2���÷�Ӧת�Ƶ��ӵ����ʵ�����9mol | |
| B�� | �÷�Ӧ��Cl2���������������ǻ�ԭ�� | |
| C�� | ���ϵ�������AgCl | |
| D�� | �μӷ�Ӧ��Cl2��$\frac{4}{9}$��ˮ��ԭ |
 �������й�PHB��˵������ȷ���ǣ�������
�������й�PHB��˵������ȷ���ǣ�������| A�� | PHB�Ǹ߷��ӻ����� | |
| B�� | �ϳ�PHB�ĵ�����CH3CH2CH��OH��COOH | |
| C�� | ͨ�����۷�Ӧ�����Ƶ�PHB | |
| D�� | ��PHB�Ľ��������һ��û�������μӷ�Ӧ |
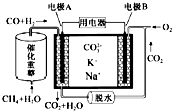
| A�� | ��ӦCH4+H2O$\frac{\underline{����}}{��}$3H2+CO��ÿ����1mol CH4ת��12mol���� | |
| B�� | ��ع���ʱ��CO32-��缫B�ƶ� | |
| C�� | �缫B�Ϸ����ĵ缫��ӦΪO2+2CO2+4e-�T2CO32- | |
| D�� | �缫A��H2����ĵ缫��ӦΪ��H2+2OH--2e-�T2H2O |

 X��Y��Z��W��N��ԭ���������������ǰ������Ԫ�أ�����X��s�ܼ���������p�ܼ���������2����Y��ԭ�Ӻ�����3��δ�ɶԵ��ӣ�Z�Ļ�̬ԭ��M����K���������ȣ�Y��Z������������֮�͵���W��������������N+ԭ�Ӻ�����3�����Ӳ��Ҹ��������ȫ��״̬���ش��������⣺
X��Y��Z��W��N��ԭ���������������ǰ������Ԫ�أ�����X��s�ܼ���������p�ܼ���������2����Y��ԭ�Ӻ�����3��δ�ɶԵ��ӣ�Z�Ļ�̬ԭ��M����K���������ȣ�Y��Z������������֮�͵���W��������������N+ԭ�Ӻ�����3�����Ӳ��Ҹ��������ȫ��״̬���ش��������⣺