��Ŀ����
���͡�������š��ɴ��Ļ��ȼ�ϳ�Һ̬˫��ˮ�⣬������һ��Һ̬���⻯�����֪�û���������Ԫ�ص���������Ϊ12�� 5%����Է�������Ϊ32���ṹ�������ָ÷��ӽṹ��ֻ�е�����
��1���õ��⻯����ĵ���Ϊ ��
��2������������Һ̬˫��ˮǡ����ȫ��Ӧ�������������ֲ���Ⱦ��������̬���ʣ�д���÷�Ӧ�Ļ�ѧ����ʽ ��
��3��NH3�����е�Nԭ����һ�Թ¶Ե��ӣ��ܷ�����Ӧ��NH3+HCl=NH4Cl����д���������⻯����ͨ����������ʱ��������Ӧ�Ļ�ѧ����ʽ
��1��N2H4�ĵ���ʽ��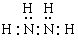 ��2��N2H4+2H2O2=N2+4H2O��3��N2H4+2HCl= N2H6Cl2��
��2��N2H4+2H2O2=N2+4H2O��3��N2H4+2HCl= N2H6Cl2��
���������������1�����ڸû��������Է�������Ϊ32����Ԫ�ص���������Ϊ12�� 5%������H��32��12�� 5%=4��N��(32-4)��14=2�����Ը����ʵķ���ʽΪN2H4�������ڸ÷��ӽṹ��ֻ�е��������������ʽΪ��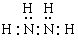 ����2��N2H4��Һ̬˫��ˮ��Ӧ�����������ֲ���Ⱦ��������̬���ʣ��÷�Ӧ�Ļ�ѧ����ʽΪN2H4+2H2O2=N2+4H2O����3���������⽫N2H4ͨ����������ʱ��������Ӧ�Ļ�ѧ����ʽΪN2H4+2HCl= N2H6Cl2��
����2��N2H4��Һ̬˫��ˮ��Ӧ�����������ֲ���Ⱦ��������̬���ʣ��÷�Ӧ�Ļ�ѧ����ʽΪN2H4+2H2O2=N2+4H2O����3���������⽫N2H4ͨ����������ʱ��������Ӧ�Ļ�ѧ����ʽΪN2H4+2HCl= N2H6Cl2��
���㣺����N2H4�ķ���ʽ���ƶϡ��ṹ�����ʵ�֪ʶ��

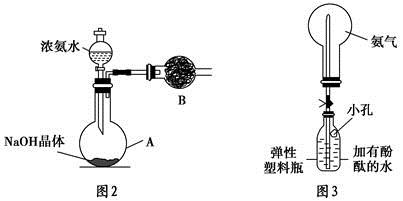
��֪KMnO4��Ũ�����ڳ����·�Ӧ�ܲ���Cl2��������ͼ��ʾ��ʵ��װ�����Ʊ��������������������������������ķ�Ӧ��ÿ�����߿��ʾһ����Ԫװ�ã������д�����ǡ��� ��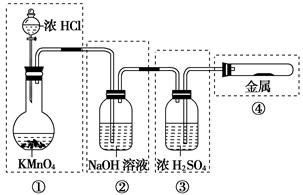
| A���ٺ͢ڴ� | B���ڴ� | C���ں͢۴� | D���ڢܴۢ� |
���ڵؿ��еĺ����ϸߡ��輰�仯����Ŀ��������Ѿã����ִ��������й㷺Ӧ�á��ش��������⣺
(1)1810����仯ѧ�ұ�������˹�ڼ���ʯӢɰ��ľ̿����ʱ���õ�һ�֡������������֡������������� ��
(2)�մɡ�ˮ��Ͳ����dz��õĹ����β��ϡ����У�������ͨ��������Ҫԭ���� ��
(3)�ߴ������ִ���Ϣ���뵼��������Ȳ�ҵ����Ҫ�Ļ������ϡ���ҵ���ᴿ���ж���·�ߣ�����һ�ֹ�������ʾ��ͼ����Ҫ��Ӧ���£�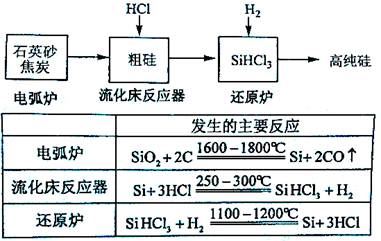
����ʯӢɰ�ͽ�̿�ڵ绡¯�и��¼���Ҳ��������̼���裬�÷�Ӧ�Ļ�ѧ����ʽΪ ��̼�����ֳ� ���侧��ṹ�� ���ơ�
������������Ӧ�IJ����У�SiHCl3��Լռ85%������SiCl4��SiH2Cl2��SiH3Cl�ȣ��й����ʵķе��������±����ᴿSiHCl3����Ҫ���ղ��������dz����������� ��
| ���� | Si | SiCl4 | SiHCl3 | SiH2Cl2 | SiH3Cl | HCl | SiH4 |
| �е�/�� | 2355 | 57.6 | 31.8 | 8.2 | -30.4 | -84.9 | -111.9 |
��SiHCl3����ˮ�⣬����ȫˮ��IJ���Ϊ ��
(4)�ȼҵ��Ϊ�������������ṩ����ԭ�ϣ���Щԭ���� ��


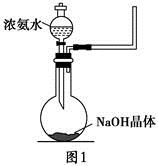

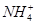 ��OH����NaOH����ʹ�ÿ��淴Ӧ��ƽ�������ƶ�
��OH����NaOH����ʹ�ÿ��淴Ӧ��ƽ�������ƶ�