��Ŀ����
����Ŀ��������M���Ʊ�һ�ֿ���ҩ���м��壬ʵ�����Է��㻯����AΪԭ���Ʊ�M��һ�ֺϳ�·�����£�
��֪��R1CH2Br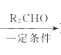 R1CH=CHR2
R1CH=CHR2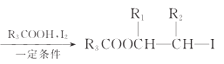
�ش��������⣺
(1)A�Ľṹ��ʽΪ_______��B�й����ŵ�����Ϊ_______��C�Ļ�ѧ����Ϊ_________��
(2)��C����D������Լ�������Ϊ_______���÷�Ӧ����Ϊ____________.
(3)��F����M�Ļ�ѧ����ʽΪ__________________________________��
(4)QΪM��ͬ���칹�壬��������������Q�Ľṹ��________��(���������칹)����д������һ.�ֺ˴Ź�����������4�����շ�Ľṹ��ʽ_________________��
�ٳ���������������״�ṹ
���ܷ���ˮ�ⷴӦ��������Ӧ
(5)���������ϳ�·�ߺ���Ϣ������ϩ����ȩΪԭ��(���Լ���ѡ)������Ʊ���2-��ϩ�ĺϳ�·��_____________��
���𰸡�![]() ȩ�� �ڼ�������(��2-��������) Br2������ ȡ����Ӧ
ȩ�� �ڼ�������(��2-��������) Br2������ ȡ����Ӧ 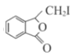 +NaOH
+NaOH![]()
![]() +NaI+H2O 4
+NaI+H2O 4 ![]() ��
��![]()
![]()
��������
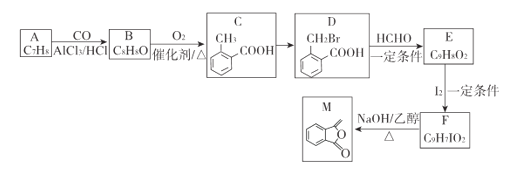
A����ʽ��C7H8��������ʷ�Ӧת��������C�Ľṹ��֪A��![]() ��A��CO��AlCl3��HCl���������·�����Ӧ����B��
��A��CO��AlCl3��HCl���������·�����Ӧ����B�� ��B����������CΪ
��B����������CΪ ��C��Br2�ڹ��������·���ȡ����Ӧ����
��C��Br2�ڹ��������·���ȡ����Ӧ���� ��D��HCHO������Ӧ����E��
��D��HCHO������Ӧ����E�� ��E��I2��һ�������·�����Ϣ�з�Ӧ����F��
��E��I2��һ�������·�����Ϣ�з�Ӧ����F�� ��F��NaOH���Ҵ���Һ���ȷ�����ȥ��Ӧ����M��
��F��NaOH���Ҵ���Һ���ȷ�����ȥ��Ӧ����M��![]() ��
��
����Ϣ��֪��AΪ![]() ��BΪ
��BΪ ��EΪ
��EΪ ��FΪ
��FΪ ��
��
(1)A�Ľṹ��ʽΪ![]() ��
�� �й����ŵ�����Ϊȩ����
�й����ŵ�����Ϊȩ���� �Ļ�ѧ����Ϊ�ڼ�������(��2-��������)��
�Ļ�ѧ����Ϊ�ڼ�������(��2-��������)��
(2) ��������������Br2����ȡ����Ӧ����
��������������Br2����ȡ����Ӧ���� ��
��
(3) ������ȥ��Ӧ����
������ȥ��Ӧ����![]() �Ļ�ѧ����ʽΪ��
�Ļ�ѧ����ʽΪ�� +NaOH
+NaOH![]()
![]() +NaI+H2O��
+NaI+H2O��
(4)MΪ![]() ������Ϣ����ͬ���칹��Q�к���HCOO-��-C��C-�����������Ľṹ��
������Ϣ����ͬ���칹��Q�к���HCOO-��-C��C-�����������Ľṹ��![]() ��
��![]() (�ڡ��䡢��λ3��)��4�֣��ٸ��ݺ˴Ź���������4�����շ壬����֪Q�Ľṹ��ʽΪ
(�ڡ��䡢��λ3��)��4�֣��ٸ��ݺ˴Ź���������4�����շ壬����֪Q�Ľṹ��ʽΪ![]() ��
��![]() ��
��
(5)��2-��ϩ�ĵ���ΪCH3CH=CHCH3���õ������CH3CH2Br��CH3CHO������Ϣ��ԭ���Ʊ���CH3CH2Br����CH2=CH2��HBr�ӳɵõ����ʺϳ�·��ΪCH2=CH2![]() CH3CH2Br
CH3CH2Br![]() CH3CH=CHCH3
CH3CH=CHCH3![]()
 ��
��

����Ŀ�������ֶ�����Ԫ�أ����ǵĽṹ�����ʵ���Ϣ���±�������
Ԫ�� | �ṹ�����ʵ���Ϣ |
A | �Ƕ������У���ϡ�������⣩ԭ�Ӱ뾶����Ԫ�أ���Ԫ�ص�ij�ֺϽ���ԭ�ӷ�Ӧ�ѵĵ��ȼ� |
B |
|
C | Ԫ�ص���̬�⻯�K������ˮ������������� |
D | �Ǻ�ˮ�г��⡢��Ԫ���⺬������Ԫ�أ��䵥�ʻ���Ҳ������ˮ���������г��õ�����ɱ���� |
����ݱ�����Ϣ��д��
��1��![]() ԭ�ӵĺ�������Ų�ʽ��___________________��
ԭ�ӵĺ�������Ų�ʽ��___________________��
��2��![]() Ԫ����Ԫ�����ڱ��е�λ��________�����Ӱ뾶��
Ԫ����Ԫ�����ڱ��е�λ��________�����Ӱ뾶��![]() ____
____![]() ����������������������
����������������������
��3��![]() ԭ�ӵĵ����Ų�ͼ��______��������ߵĵ���Ϊ______����ϵĵ��ӣ�������______�Ρ�
ԭ�ӵĵ����Ų�ͼ��______��������ߵĵ���Ϊ______����ϵĵ��ӣ�������______�Ρ�
��4��![]() �Ľṹʾ��ͼ��________________��
�Ľṹʾ��ͼ��________________��
��5��![]() ������������Ӧ��ˮ������
������������Ӧ��ˮ������![]() ������������Ӧ��ˮ���ﷴӦ�Ļ�ѧ����ʽΪ________����
������������Ӧ��ˮ���ﷴӦ�Ļ�ѧ����ʽΪ________����![]() ����������Ӧ��ˮ���ﷴӦ�Ļ�ѧ����ʽΪ________________��
����������Ӧ��ˮ���ﷴӦ�Ļ�ѧ����ʽΪ________________��
��6����һʵ��˵��![]() Ԫ�صķǽ����Ա�
Ԫ�صķǽ����Ա�![]() Ԫ�ص�ǿ_______________
Ԫ�ص�ǿ_______________
����Ŀ����1���������Ļ�����Ӧ�ù㷺����FeCl3������������ӡˢ��·ͭ�帯ʴ��������ֹѪ���ȡ�д��FeCl3��Һ��ʴӡˢ��·ͭ������ӷ���ʽ___����ʴͭ���Ļ����Һ�У���Cu2+��Fe3+��Fe2+��Ũ�Ⱦ�Ϊ0.10mol/L��������±����������ݺ�ҩƷ��������ȥCuCl2��Һ��Fe3+��Fe2+��ʵ�鲽��___��
�������↑ʼ����ʱ��pH | �������������ȫʱ��pH | |
Fe3+ Fe2+ Cu2+ | 1.9 7.0 4.7 | 3.2 9.0 6.7 |
�ṩ��ҩƷ��Cl2 ŨH2SO4 NaOH��Һ CuO Cu | ||
��2����ӵ绯ѧ���ɶԴ�����Ⱦ��NO���������������乤��ԭ����ͼ��ʾ������Ĥ����H+��H2Oͨ�����缫��Ϊ___�����缫��ӦʽΪ___���������еķ�ӦΪ____��ÿ����1molNO����ͬʱ�õ�___gO2��
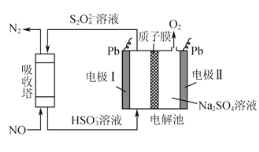
��3��������-����-����̼������ȼ�ϵ��װ����ͼ��ʾ������˵�����������______��
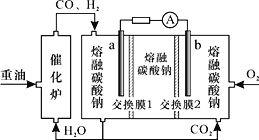
A.O2��b���õ��ӣ����ձ���ԭΪCO32-
B.�ŵ�����У������ɵ缫a����������缫b
C.�õ�ع���ʱ��CO32-��������Ĥ2������b��
D.H2����ĵ缫��ӦΪH2-2e-+CO32-=H2O+CO2