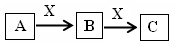��Ŀ����
��֪A��B��C����ѧ��ѧ�ij������ʣ�������һ��������������ת����ϵ��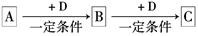
��1����A��ʹʪ��ĺ�ɫʯ����ֽ������CΪ����ɫ���塣��Aת��ΪB��Ӧ�Ļ�ѧ����ʽΪ ��
��2����D�ǽ�����C��Һ������ʱӦ��������D���������ǣ��ñ�Ҫ�����ֺ����ӷ���ʽ˵���� ��
��3����D��һ�ֳ������������壬A��һ��ǿ���������ˮ��Һ�е���������������Ӿ�����10�����ӣ�����C���ȷֽ�ɵù���B���÷�Ӧ�Ļ�ѧ����ʽ�� ��
��4����DΪ�ȼҵ����Ҫ��Ʒ��B�������ԣ���Bת��ΪC�����ӷ���ʽΪ ��
��5����A��B��C��Ϊ�����D��һ�ֺ�ɫ��̬�ǽ������ʣ���Ԫ��D�����ڱ��е�λ
���� ��B���ӵĽṹʽΪ ��
��1��4NH3 + 5O2 4NO + 6H2O��2����ֹFe2+��������2F3+ + Fe = 3Fe2+��3��2NaHCO3
4NO + 6H2O��2����ֹFe2+��������2F3+ + Fe = 3Fe2+��3��2NaHCO3 Na2CO3 + H2O +CO2����4��Al(OH)3 + OH- = AlO2- + 2H2O��5����2���ڡ��ڢ�A�壬O=C=O
Na2CO3 + H2O +CO2����4��Al(OH)3 + OH- = AlO2- + 2H2O��5����2���ڡ��ڢ�A�壬O=C=O
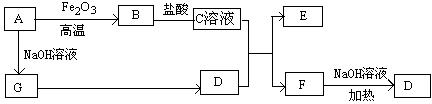
�������������A��D��Ӧ����B��B��D��Ӧ����C����B��C�к��е�ij�ֹ�ͬԪ���DZ��Ԫ�ء�
��1����A��ʹʪ��ĺ�ɫʯ����ֽ��������A��NH3��CΪ����ɫ����NO2��NH3��D�ܷ�Ӧ����B����B��NO��D��O2��Aת��ΪB�ķ�Ӧ����ʽΪ4NH3+5O2 4NO+6H2O��2��D�ǽ�����C��Һ������ʱӦ��������D��˵��C�ױ����������ʣ���AΪCl2��BΪFeCl3��CΪFeCl2��DΪFe��������������Һ�м�������ɷ�ֹFe2+�����������ӷ���ʽ2F3+ + Fe = 3Fe2+��3����D��һ�ֳ������������壬��D�Ƕ�����̼��A��һ��ǿ���������ˮ��Һ�е���������������Ӿ�����10�����ӣ���A��NaOH����BΪNa2CO3��CΪNaHCO3��Bת��ΪC�����ӷ���ʽΪCO2+CO32-+H2O=2HCO3-��4��DΪ�ȼҵ����Ҫ��ƷΪNaOH��B���������ƶ�ΪAl(OH)3����C��Һ��Al(OH)3��NaOH��Ӧ����NaAlO2�����ӷ���ʽAl(OH)3 + OH- = AlO2- + 2H2O��5����A��B��C��Ϊ�����D��һ�ֺ�ɫ��̬�ǽ������ʣ��ƶ�ΪDΪC��BΪCO2��CΪCO����Ԫ��D�����ڱ��е�λ���ǵ�2���ڡ���IVA�壬B���ӵĽṹʽΪO=C=O��
4NO+6H2O��2��D�ǽ�����C��Һ������ʱӦ��������D��˵��C�ױ����������ʣ���AΪCl2��BΪFeCl3��CΪFeCl2��DΪFe��������������Һ�м�������ɷ�ֹFe2+�����������ӷ���ʽ2F3+ + Fe = 3Fe2+��3����D��һ�ֳ������������壬��D�Ƕ�����̼��A��һ��ǿ���������ˮ��Һ�е���������������Ӿ�����10�����ӣ���A��NaOH����BΪNa2CO3��CΪNaHCO3��Bת��ΪC�����ӷ���ʽΪCO2+CO32-+H2O=2HCO3-��4��DΪ�ȼҵ����Ҫ��ƷΪNaOH��B���������ƶ�ΪAl(OH)3����C��Һ��Al(OH)3��NaOH��Ӧ����NaAlO2�����ӷ���ʽAl(OH)3 + OH- = AlO2- + 2H2O��5����A��B��C��Ϊ�����D��һ�ֺ�ɫ��̬�ǽ������ʣ��ƶ�ΪDΪC��BΪCO2��CΪCO����Ԫ��D�����ڱ��е�λ���ǵ�2���ڡ���IVA�壬B���ӵĽṹʽΪO=C=O��
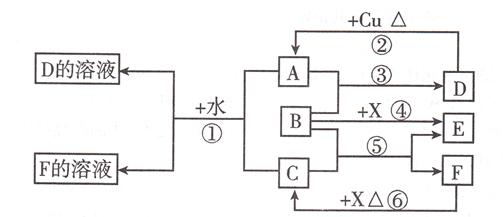
���㣺������ƶ�

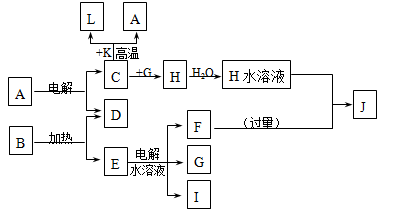
��֪A��F���ֳ��������У�B�ǿ�������Ҫ�ɷ�֮һ���������dz����Ļ��������E��һ�ֳ������ܼ���F��һ���������塣����֮���ת����ϵ��ͼ��ʾ������˵������ȷ���� ����������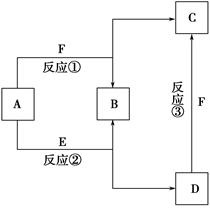
| A��������A��һ�ּ��������� |
| B��ͼ�Т٢ڢ�������Ӧ��Ϊ������ԭ��Ӧ |
| C��������D��һ��ǿ�� |
| D��������C��ˮ��Һ������ |