��Ŀ����
�輰�仯����������ִ�������������ף���ش������й����⣺
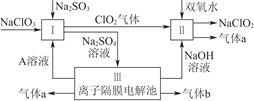
(1)��ԭ�ӵĽṹʾ��ͼ��________��
(2)������Ʒ���豸���õIJ������ڹ����ε���________��
�ٳ�����Ͽˮ���ӣ���ʯӢ���ά�����մ�����������ͨ�������ݹ�̫���ܵ��
A���٢ڢ� B���ۢܢ� C���ڢۢ� D���٢ۢ�
(3)�����£�SiCl4ΪҺ̬���е�Ϊ57.6�棬�ڿ�����ð�������Ʊ��ߴ��ȹ���м����SiCl4������Һ̬���ʣ���Ҫ�õ��ߴ���SiCl4��Ӧ���õķ�����________���û�ѧ����ʽ����Ҫ���ֽ���SiCl4�ڿ�����ð������ԭ��_______________________________________��
(4)��ҵ�Ͽ���SiCl4(g)�Ʊ����½ṹ�մɵ����裬�䷴Ӧ����ʽΪ
3SiCl4(g)��2N2(g)��6H2(g) Si3N4(s)��12HCl(g)����H��a kJ/mol(a��0)
Si3N4(s)��12HCl(g)����H��a kJ/mol(a��0)
�ٸ÷�Ӧ��ƽ�ⳣ������ʽK��______________.
�����ܱպ��������У��ܱ�ʾ������Ӧ�ﵽƽ��״̬����________��
A��3v��(N2)��v��(H2)
B��v��(HCl��4v����4v��(SiCl4)
C����������ܶȱ��ֲ���
D��c(N2)��c(H2)��c(HCl)��1��3��6
����ij�����´ﵽƽ��ʱ��H2��HCl���ʵ���֮��Ϊm��n�����������������䣬�����¶ȴﵽƽ��ʱ��H2��HCl���ʵ���֮��________m��n(�>��������������)��
(1)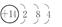
(2)D
(3)��������������SiCl4��3H2O=H2SiO3��4HCl��HCl��ˮ������������
(4)��K��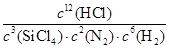 ����AC����<
����AC����<
����

��γ�ȥ���и���ĩ״������е�����(������Ϊ����)����ѡ�������ṩ���Լ��Ͳ�������������ڱ��ڡ�
��ѡ�Լ���A���B�ռ���Һ��C������Dˮ��ECO2��F���������Լ�
��ѡ�������ټ��ȣ��ڼ������ڣ��۹��ˣ��ܽᾧ
| �����ʵ����� | �����Լ� | ��Ҫ���� |
| (1)SiO2(NaHCO3) | | |
| (2)SiO2(CaCO3) | | |
| (3)SiO2(Si) | | |
| (4)NaCl(SiO2) | | |
(1)�����뺬�Ȼ������йص�˵����ȷ����________(����ĸ)��
| A��HClO�����ᣬ����NaClO��������� |
| B�����ˮ����μ�����������FeCl3��Һ�����Ƶ�Fe(OH)3���� |
| C��HCl��Һ��NaCl��Һ��ͨ�����ӵ��磬����HCl��NaCl�������ӻ����� |
| D�����NaCl��Һ�õ�22.4 L H2(��״��)����������Ҫת��NA������(NA��ʾ�����ӵ�����) |
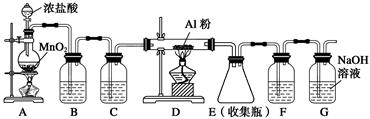
װ��B��ʢ�ű���NaCl��Һ����װ�õ���Ҫ������______________��F���Լ���������____________________����һ������װ���ʵ��Լ����ͬʱ��F��G�����ã���װ����Լ�Ϊ________________��
���꣬Ϊ����������ʣ�����������������Ϊ��߾���Ч�棬��������������ܶ�ܻ����������̽�����ٽ���ѧ��ҵ�ķ�չ��
��1�����ڹ������̵�Ӧ�ã����÷��糧������SO2�Ƴ��Է���أ����ط�Ӧ����ʽΪ��2SO2��O2��2H2O��2H2SO4���õ�ص綯��Ϊ1.06V��ʵ�ʹ����У���SO2ͨ���ص� �����������������������ӦʽΪ �������ַ�������SO2�������ŵ��� ��
��2�������Ṥҵ��SO2β������ˮ��ʯ��ʯ����̿��̼����狀��Ȼ��ص�Ϊԭ�ϣ����Ժϳ�����ҪӦ�ü�ֵ���ơ�����ء�������淋����ʣ��ϳ�·�����£�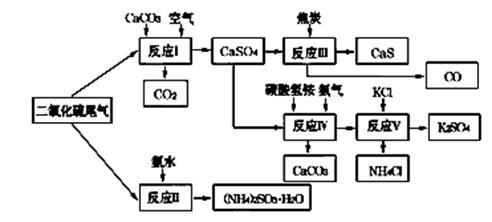
�������У���Ӧ���е���Һ�м���������ԭ�Ժ�ǿ�ĶԱ����ӵ����ʣ���Ŀ���� ��
�������й�˵����ȷ���� (�����)��
| A����Ӧ��������������������Ա�֤����������������������� |
B����Ӧ���з�����Ӧ�Ļ�ѧ����ʽΪCaSO4��4C  CaS��4CO�� CaS��4CO�� |
| C����Ӧ���������60��70�棬Ŀ��֮һ�Ǽ���̼����淋ķֽ� |
| D����Ӧ���еĸ������Ȼ�刺��������� |
�ܣ�NH4)2SO3�����ڵ糧���̵������ѵ�������������ת��Ϊ������ͬʱ����һ�ֵ��ʣ��γɹ���ϵͳ��д������������������立�Ӧ�Ļ�ѧ����ʽ ��
Na2SO3�dz��õĿ�������
��1��ʵ����ͨ����Ũ���ᣨ1��1����Na2SO3���Ʊ�SO2���壬
��Ӧ����ʽΪ�� ���Ʊ���SO2������ͨ������ˮ���������и�����ܸ���SO2������ǣ� ��
A.Ũ���� B.��ʯ�� C.��ˮCaCl2
��2�� ����SO2����ͨ��NaOH��Һ�пɵ�NaOH��Na2SO3�Ļ����Һ����û����Һ�м���������ˮ������Һ��Ϊ��ɫ��������Һ��Br2��Na2SO3����������ԭ��Ӧ����Ӧ�����ӷ���ʽΪ______________��
��3����Ӧ�����Һ����SO32����SO42����Br����OH���������ӣ�����д��������SO32����SO42����Br����ʵ�鱨�棻
��ѡ�Լ���2 mol��L��1HCl��1 mol��L��1H2SO4��1mol��L��1HNO3��1 mol��L��1BaCl2��
1 mol��L��1Ba(NO3)2��0.1 mol��L��1AgNO3��CCl4���������Ʊ�����ˮ�����Ʊ�����ˮ��
| ��� | ʵ����� | Ԥ������ͽ��� |
| ����� | ȡ��������Һ���Թ�A�У��μ�2 mol��L��1HCl����Һ�����ԣ����뼸��________(���Լ�)���� | ________��֤������Һ�к�SO32- |
| ����� | ��ȡ��������Һ���Թ�B�У����� ���ٵμ����� 1 mol��L��1 BaCl2��Һ | |
| ����� | ��ȡ��������Һ���Թ�C�У� �������ú�۲���ɫ | ��Һ�ֲ㣬�ϲ�Һ��ʳȺ�ɫ��֤������Һ�к�Br- |
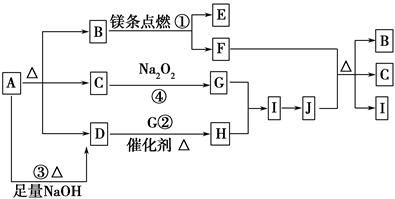
 2MgO��C������д���¿հף�
2MgO��C������д���¿հף�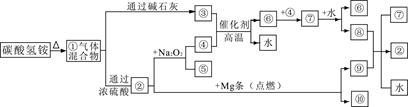
 Na2S2O3
Na2S2O3
 2NaI+Na2S4O6����Ʒ�е�Na2S2O3��5H2O�Ĵ���Ϊ��������%��
2NaI+Na2S4O6����Ʒ�е�Na2S2O3��5H2O�Ĵ���Ϊ��������%��