��Ŀ����
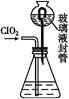
�������ƣ�NaClO2����һ��ǿ������Ư�����㷺���ڷ�֯��ӡȾ��ʳƷ��ҵ�����ڼ��Ի������ȶ����ڡ�ijͬѧ�������Ϻ��������NaClO2����Ҫ�������¡�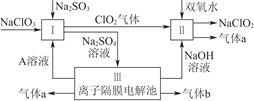
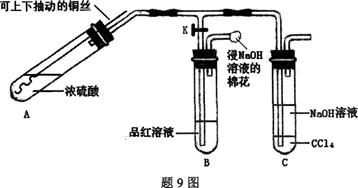
��1��˫��ˮ�ĽṹʽΪ____________�����з�����Ӧ�Ļ�ԭ����__________���ѧʽ����
��2�����з�Ӧ�����ӷ���ʽ��_____________________________________________________________________________________________________________________________________��
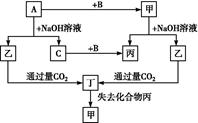
��3��A�Ļ�ѧʽ��________��װ�â���A��________����������
��4��ClO2��һ�ָ�Чˮ�������������������ƺ�ϡ����Ϊԭ���Ʊ���
��д���÷�Ӧ�Ļ�ѧ����ʽ��________________________________________________________________________________________________________________________________________________��
���о�����������Ӧ��ʼʱ����Ũ�Ƚϴ��������������Cl2�������ӷ���ʽ���Ͳ���Cl2��ԭ��________________________________________________________________________
________________________________________________________________________��
��5��NaClO2���ʿɷֽ�ΪNaClO3��NaCl��ȡ������NaClO2������һ�ݸ����ʵ�����ʹ֮���ʣ���һ���ϸ棬�������Һ�����ֱ�������FeSO4��Һ��Ӧʱ������Fe2�������ʵ���________�����ͬ��������ͬ�������жϡ�����
��1��H��O��O��H��Na2SO3
��2��2ClO2 ��H2O2��2OH��=2ClO2-��O2����2H2O
��3��H2SO4����
��4��5NaClO2��4HCl=5NaCl��4ClO2����2H2O ClO2-��3Cl����4H��=2Cl2����2H2O
��5����ͬ
����

Ӳ�ʲ������ǻ�ѧʵ���о���ʹ�õ�һ�����������������ʵ�飨�̶�װ���ԣ����ش����⡣
��Ӳ�ʲ����ܳ���Ϊ������巴Ӧ�ij�������ͼ������װ��ʡ�ԣ���Ҫʱ�ɼ��ȣ���
����ʵ����������ȷ����
| A������AΪ��ˮ����ͭʱ�����Լ�������X���Ƿ���ˮ���� |
| B������AΪͭʱ�����Գ�ȥN2�е�O2 |
| C���ڿ����������£������Ȼ�������õ���������ˮ�Ȼ��� |
| D���������Ϳ��������ͨ�����ȵIJ�˿���ɿ����к���ɫ�������� |
a��b��c��������仯���±�������д���еĿհף�
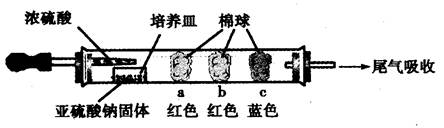
| ���� | �����ϵμӵ��Լ� | ʵ������ | ���ͺͽ��� |
| a | | �����ף��Ⱥ��ָֻ���ɫ | |
| b | ����̪��NaOH��Һ | �����Ϊ��ɫ | ���ӷ���ʽ�� |
| c | | �����Ϊ��ɫ | ���ۣ���������� �� |
 SiHCl3��H2��
SiHCl3��H2��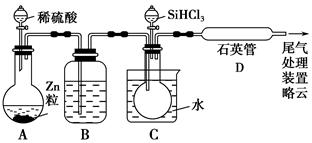
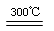
 Si3N4(s)��12HCl(g)����H��a kJ/mol(a��0)
Si3N4(s)��12HCl(g)����H��a kJ/mol(a��0)
 2I-+S4
2I-+S4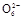 ),����ȥV mL�����������Һ��
),����ȥV mL�����������Һ��