��Ŀ����
��5�֣����������仯�����ںϽ�����Լ������ȷ���Ӧ�ù㷺����ش��������⣺
��1��Niԭ�ӵĻ�̬�����Ų�ʽΪ______________________________��
��2��NiO��FeO�ľ���ṹ���;����Ȼ��Ƶ���ͬ��Ni2+��Fe2+�����Ӱ뾶�ֱ�Ϊ69 pm��78 pm�����۵�NiO ________ FeO(�<����>��)��
��3������������(La)�γɵĺϽ���һ�����õĴ�����ϣ��侧���ṹʾ��ͼ������ͼʾ���úϽ�Ļ�ѧʽΪ_______________��

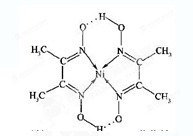
��4������ͪ뿳����ڼ���Ni2+����ϡ��ˮ�����У�����ͪ���Ni2+��Ӧ�������ʺ�ɫ��������ṹ������ͼ��ʾ���ýṹ�У���ԭ�ӵ��ӻ��������Ϊ_______________����ԭ�ӵ��ӻ��������Ϊ_______________��
��1��Niԭ�ӵĻ�̬�����Ų�ʽΪ______________________________��
��2��NiO��FeO�ľ���ṹ���;����Ȼ��Ƶ���ͬ��Ni2+��Fe2+�����Ӱ뾶�ֱ�Ϊ69 pm��78 pm�����۵�NiO ________ FeO(�<����>��)��
��3������������(La)�γɵĺϽ���һ�����õĴ�����ϣ��侧���ṹʾ��ͼ������ͼʾ���úϽ�Ļ�ѧʽΪ_______________��

��4������ͪ뿳����ڼ���Ni2+����ϡ��ˮ�����У�����ͪ���Ni2+��Ӧ�������ʺ�ɫ��������ṹ������ͼ��ʾ���ýṹ�У���ԭ�ӵ��ӻ��������Ϊ_______________����ԭ�ӵ��ӻ��������Ϊ_______________��
��1��1s22s22p63s23p63d84s2 ��2���� ��3��LaNi5 ��4��sp2 sp3
��1��Niԭ�ӵ���������28�����Ը��ݹ���ԭ����֪��Ni�Ļ�̬�����Ų�ʽΪ1s22s22p63s23p63d84s2��
��2��NiO��FeO�ľ���ṹ���;����Ȼ��Ƶ���ͬ����˶������Ӿ��塣����Ni2+��Fe2+�����Ӱ뾶�ֱ�Ϊ69 pm��78 pm������NiO�����Ӽ�ǿ��FeO�����Ӽ�������ǰ�ߵ��۵���ں��ߡ�
��3�����ݾ����ṹ��֪�����е�Laԭ����8��1/8��1��Niԭ����8��1/2��1��5�����Ի�ѧʽΪLaNi5��
��4�����ݽṹ��֪����ԭ���γ�1��˫����1��������������sp2�ӻ�����ԭ��ȫ���ǵ����������sp3�ӻ���
��2��NiO��FeO�ľ���ṹ���;����Ȼ��Ƶ���ͬ����˶������Ӿ��塣����Ni2+��Fe2+�����Ӱ뾶�ֱ�Ϊ69 pm��78 pm������NiO�����Ӽ�ǿ��FeO�����Ӽ�������ǰ�ߵ��۵���ں��ߡ�
��3�����ݾ����ṹ��֪�����е�Laԭ����8��1/8��1��Niԭ����8��1/2��1��5�����Ի�ѧʽΪLaNi5��
��4�����ݽṹ��֪����ԭ���γ�1��˫����1��������������sp2�ӻ�����ԭ��ȫ���ǵ����������sp3�ӻ���

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ

 ���������к��еĻ�ѧ�������� ������ĸ��ţ���
���������к��еĻ�ѧ�������� ������ĸ��ţ��� ��E+δ������������ɫ�����һ��������E+�ĸ���Ϊ ����
��E+δ������������ɫ�����һ��������E+�ĸ���Ϊ ����