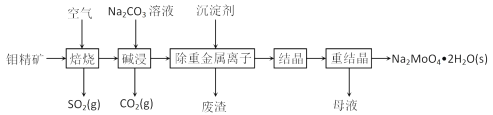
��Ŀ����
����Ŀ���ﶬ���ҹ������ֵ�������������������������������PM2.5ϸ���Ӱ���(NH4)2SO4��NH4NO3���л������P�ﳾ�ȣ����������л�����ͭ���ؽ���Ԫ�ء�
(1)PM2.5�����������ж����к����ʣ����������ι⻯ѧ�������⻯ѧ�����к���NOx��HCOOH��![]() �ȶ�����Ⱦ�
�ȶ�����Ⱦ�
����̬Nԭ�ӵĵ����Ų�ʽ___________��C��N��O�ĵ�һ�������ɴ�С��˳��Ϊ___________
�������飨NH3BH3������Ϊ�����DZ�������ʹ������֮һ�������д�����λ�����ṩ�µ��ӶԵijɼ�ԭ����________��д��һ���백���黥Ϊ�ȵ�����ķ���________���ѧʽ����
(2)PM2.5ϸ���Ӱ���(NH4)2SO4��NH4NO3�ȡ�
��(NH4)2SO4�����и���������ڵ���������________(�����)��
a�����Ӽ���b�����ۼ���c����λ����d�����»�����
��NH4NO3�������ӵĿռ乹��Ϊ________�������ӵ�����ԭ�ӹ������________�ӻ���
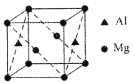
(3)ij��þ���Ͻ����Ϊ���Ʋ��ϣ��úϽ��ṹ��ͼ��ʾ�������ⳤΪanm���úϽ�Ļ�ѧʽΪ____________��������ÿ��þԭ����Χ�����������ԭ����ĿΪ___________���þ�����ܶ�Ϊ__________________g/cm3(����٤����������ֵ��NA��ʾ)��
���𰸡�1s22s22p3 N>O>C N C2H6 abc ƽ�������� sp2 Mg2Al 4 ![]()
��������
(1)��Nԭ�ӵ�ԭ�ӽṹʾ��ͼΪ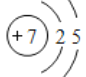 ��ͬ������ԭ�����������һ�����ܳ��������ƣ�NԪ��ԭ��2p �ܼ�Ϊ������ȶ�״̬����һ�����ܸ���ͬ��������Ԫ�صģ�
��ͬ������ԭ�����������һ�����ܳ��������ƣ�NԪ��ԭ��2p �ܼ�Ϊ������ȶ�״̬����һ�����ܸ���ͬ��������Ԫ�صģ�
��Bԭ���пչ����NH3��Nԭ����1�Թµ��Ӷԣ��백���黥Ϊ�ȵ�����ķ��ӣ�������2��Cԭ�Ӵ���B��Nԭ�ӣ�
(2)��(NH4)2SO4������笠����Ӻ����������֮�������Ӽ�����笠����Ӻ�����������ڲ����ǹ��ۼ���
��NH4+������ԭ�ӵ�ԭ�Ӽ۲���Ӷ���=4+![]() =4���µ��ӶԸ�����0��NO3-������ԭ�ӵ�ԭ�Ӽ۲���Ӷ���=3+
=4���µ��ӶԸ�����0��NO3-������ԭ�ӵ�ԭ�Ӽ۲���Ӷ���=3+![]() =3�����ݼ۲���ӶԻ��������ж����ӿռ乹�ͼ�����ԭ���ӻ���ʽ��
=3�����ݼ۲���ӶԻ��������ж����ӿռ乹�ͼ�����ԭ���ӻ���ʽ��
(3) �þ�����Alԭ�Ӹ���Ϊ2��![]() =1��Mgԭ�Ӹ���=8��
=1��Mgԭ�Ӹ���=8��![]() +2��
+2��![]() =2��������ÿ��þԭ����Χ�����������ԭ����ĿΪ4���������=(a��10-7 cm)3���þ�����ܶ�=
=2��������ÿ��þԭ����Χ�����������ԭ����ĿΪ4���������=(a��10-7 cm)3���þ�����ܶ�=![]() ��
��
(1)����Nԭ�ӵ�ԭ�ӽṹʾ��ͼ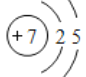 ��֪��̬Nԭ�ӵĵ����Ų�ʽΪ1s22s22p3��ͬ������ԭ�����������һ�����ܳ��������ƣ�NԪ��ԭ��2p �ܼ�Ϊ������ȶ�״̬����һ�����ܸ���ͬ��������Ԫ�صģ���һ�������ɴ�С��˳��Ϊ��N��O��C��
��֪��̬Nԭ�ӵĵ����Ų�ʽΪ1s22s22p3��ͬ������ԭ�����������һ�����ܳ��������ƣ�NԪ��ԭ��2p �ܼ�Ϊ������ȶ�״̬����һ�����ܸ���ͬ��������Ԫ�صģ���һ�������ɴ�С��˳��Ϊ��N��O��C��
��Bԭ���пչ����NH3��Nԭ����1�Թµ��Ӷԣ�Nԭ���ṩ�µ��ӶԸ�Bԭ���γ���λ�����백���黥Ϊ�ȵ�����ķ��ӣ�������2��Cԭ�Ӵ���B��Nԭ�ӣ��백����ȵ�����һ�ַ���Ϊ��CH3CH3����ѧʽΪC2H6��
(2)��(NH4)2SO4������笠����Ӻ����������֮�������Ӽ�����笠����Ӻ�����������ڲ����ǹ��ۼ�������һ��Hԭ�Ӻ�Nԭ���γ���λ����������в����ڵ�������Ϊ���»������ʴ�Ϊabc��
��NH4+������ԭ�ӵ�ԭ�Ӽ۲���Ӷ���=4+![]() =4���µ��ӶԸ�����0���乹��Ϊƽ����������NO3-������ԭ�ӵ�ԭ�Ӽ۲���Ӷ���=3+
=4���µ��ӶԸ�����0���乹��Ϊƽ����������NO3-������ԭ�ӵ�ԭ�Ӽ۲���Ӷ���=3+![]() =3�����ݼ۲���ӶԻ�������֪笠����ӿռ乹������������ṹ���������������ԭ���ӻ���ʽΪsp2��
=3�����ݼ۲���ӶԻ�������֪笠����ӿռ乹������������ṹ���������������ԭ���ӻ���ʽΪsp2��
(3) �þ�����Alԭ�Ӹ���Ϊ2��![]() =1��Mgԭ�Ӹ���=8��
=1��Mgԭ�Ӹ���=8��![]() +2��
+2��![]() =2�������仯ѧʽΪMg2Al��������ÿ��þԭ����Χ�����������ԭ����ĿΪ4���������=(a��10-7cm)3���þ�����ܶ�=
=2�������仯ѧʽΪMg2Al��������ÿ��þԭ����Χ�����������ԭ����ĿΪ4���������=(a��10-7cm)3���þ�����ܶ�=![]() =
= g/cm3=
g/cm3=![]() g/cm3��
g/cm3��