��Ŀ����
����Ŀ����һ���Ϊ10L�������У�ͨ��һ������CO��H2O(g)����850��ʱ�������·�Ӧ��CO��g�� + H2O��g�� ![]() CO2��g�� + H2��g����H��0��CO��H2O(g)Ũ�ȱ仯����ͼ
CO2��g�� + H2��g����H��0��CO��H2O(g)Ũ�ȱ仯����ͼ
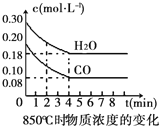
��1��0��4min��ƽ����Ӧ����v��CO��=___________molL-1min-1��
��2����������������Ӧ��ƽ�ⳣ��K=___________��д����ʽ����
��3���÷�Ӧƽ��ʱCO��ת����Ϊ________________��
��4�����жϸ÷�Ӧ�ﵽ��ѧƽ��״̬��������______________��
A��������ѹǿ���� B�����������c(CO)����
C��v(H2)��=v(H2O)�� D��c(CO2)=c(CO)
��5��t��������850����ʱ������ͬ�����з���������Ӧ�������ڸ����ʵ�Ũ�ȱ仯���±���

�� c1��ֵ___________0.08molL-1������ڡ�С�ڻ���ڣ���
�ڷ�Ӧ��4min��5min�䣬ƽ�����淽���ƶ������ܵ�ԭ����________________������5min��6min֮����ֵ�����仯�����ܵ�ԭ����_______________��
a������ˮ���� b�������¶� c��ʹ�ô��� d����������Ũ��
���𰸡���1��0.03��2�֣�����2��![]() ��2�֣���
��2�֣���
��3��60%��2�֣�����4��BC��2�֣���
��5����>��2�֣�����d��a����1�֣�
��������
�����������1������ͼ���֪����0��4min��CO��Ũ�ȼ�С��0.20mol/L��0.08mol/L��0.12mol/L�������䷴Ӧ������0.12mol/L��4min��0.03mol/��L��min����
��2����ѧƽ�ⳣ������һ�������£������淴Ӧ�ﵽƽ��״̬ʱ��������Ũ�ȵ���֮���ͷ�Ӧ��Ũ�ȵ���֮���ı�ֵ�����Ը��ݷ���ʽ��֪ƽ�ⳣ������ʽΪ![]() ��
��
��3���÷�Ӧƽ��ʱCO��ת����Ϊ![]() ��
��
��4��A����Ӧǰ��������䣬������ѹǿ���䲻��˵����Ӧ�ﵽƽ��״̬��A����B�����������c(CO)����˵����Ӧ�ﵽƽ��״̬��B��ȷ��C��v(H2)��=v(H2O)����ʾ���淴Ӧ������ȣ�˵����Ӧ�ﵽƽ��״̬��C��ȷ��D��c(CO2)=c(CO)û��˵����Ӧ���ʵķ�����һ���ﵽƽ��״̬��D����ѡBC��
��5���ٸ��ݱ������ݿ�֪���ڴ�ʱ���ڣ����ʵ�Ũ���Dz���ģ����Է�Ӧ����ƽ��״̬�����ڷ�Ӧ�Ƿ��ȷ�Ӧ�����������¶ȣ�ƽ�����淴Ӧ�����ƶ������CO��Ũ�ȴ���0.08mol/L��
������Ӧ��Ũ�Ȼ��¶ȣ�ƽ��������Ӧ�����ƶ������������ܸı�ƽ��״̬������Ӧ��������������Ũ�ȣ�ƽ�����淴Ӧ�����ƶ�����ѡd�����ݱ������ݿ�֪��5min��6min��CO��Ũ�ȼ�С����ˮ������������Ũ������˵���ı��������������ˮ������Ũ�ȣ�ƽ��������Ӧ�����ƶ�����ѡa��

 ��������ϵ�д�
��������ϵ�д�����Ŀ���״���һ����Ҫ�Ļ���ԭ�ϣ�����һ�ֿ�������Դ�����п�����Ӧ�õĹ���ǰ����
��1����֪��CH3OH(g)=HCHO(g)+H2(g) ��H=+84kJ/mol
2H2(g)+O2(g)=2H2O(g) ��H=-484kJ/mol
����ҵ�ϳ��Լ״�Ϊԭ����ȡ��ȩ����д��CH3OH(g)��O2(g)��Ӧ����HCHO(g)��H2O(g)���Ȼ�ѧ����ʽ��________________��
���������Ʊ���ȩʱ������Ӧ����ͨ���ʵ���������������Ŀ����_________________��
��2����ҵ�Ͽ������·����ϳɼ�ȩ����ѧ����ʽΪCO(g)+2H2(g)=CH3OH(g)����֪ijЩ��ѧ���ļ����������±���
��ѧ�� | C-C | C-H | H-H | C-O | C=O | O-H |
����/kJ/mol | 348 | 413 | 436 | 358 | x | 463 |
��ش��������⣺
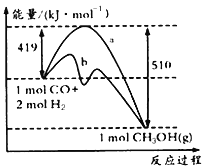
����ͼ������a������b�Ĵ�ʩ��_______________��
����֪CO�е�C��O֮��Ϊ�����������ΪxkJ/mol����x=___________��
��3���ɼ״���������NaOH��Һ���ɵ������ֻ���أ���ʹ�ֻ�����ʹ��һ���²ų�һ�ε硣
���õ�������ĵ缫��ӦʽΪ____________________��
�����Ըõ��Ϊ��Դ����ʯī���缫���200mL�����������ӵ���Һ��
���� | Cu2+ | H+ | Cl- | SO42- |
c/mol/L | 0.5 | 2 | 2 | 0.5 |
���һ��ʱ��������ռ�����ͬ�������ͬ�����£�������ʱ��������Һ����ı仯���缫������ܴ��ڵ���Һ�����������ռ�������������Ϊ_____________��
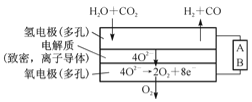
��4�����ˮ������CO2�����ϳ�����H2+CO�����ϸ��¶��£�70-1000��������SOEC����缫��ʩ��һ����ֱ����ѹ��H2O��CO2����缫������ԭ��Ӧ����O2-��O2-�������ܵĹ������������ʲ㵽�����缫�������缫����������Ӧ�õ���O2������ͼ��֪AΪֱ����Դ�� (��������������������)����д����H2OΪԭ������H2�ĵ缫��Ӧʽ��____________��
����Ŀ��
�ɷֵ�����/g | Ħ������/��gmol��1�� | |
���� | 25.00 | 342 |
����� | 0.87 | 174 |
��˾ƥ�� | 0.17 | 180 |
������� | 0.316 | 158 |
������ | 0.02 | 170 |
��1���������ʻ����ʼ�����K������˾ƥ���в���K+�������ʵ���Ũ��Ϊ__________molL������Ҫ����ԭʼ����д������ʽ���ɣ�����Ҫ��������㣩��
��2�������������ʻ����ʼ���������������ձ�����������ҩ�ס�������ƽ�������룩��
____________________������ȱ���������ƣ���
��3������Һ���ƹ����У����в��������ƽ��û��Ӱ�����_________������ĸ����
A������ʱ����������ƿ�̶���
B������ƿ��ʹ��ǰδ�����������������ˮ
C������ƿ��ʹ��ǰ�ո�������һ�����ʵ���Ũ�ȵ�NaCl��Һ��δϴ��
D������ҡ�Ⱥ���Һ���������ƿ�Ŀ̶��ߣ���δ���κδ���
��4����Ҫ����0.5mol��L��1500ml��������Һ����
��������������Ϊ98%���ܶ�Ϊ1.84g��cm��3��Ũ��������Ϊ___________������������һλС����mL;
�����ʵ������15mL��20mL��50mL��Ͳ��Ӧѡ��___________mL��Ͳ���;
�����ƹ������������ձ��н�Ũ����ϡ�ͣ�ϡ��ʱ����������_____________________.