��Ŀ����
A��B��C����Ԫ�ص�ԭ�Ӿ�����ͬ�ĵ��Ӳ�������B�ĺ˵������A��1��Cԭ �ӵĵ���������Bԭ�ӵĵ���������4�� 1molA�ĵ��ʸ��������ᷴӦ�����û�����״����22.4L��H2����ʱAת��Ϊ����ԭ�Ӿ�����ͬ���Ӳ�ṹ�����ӡ���ش�
��1���õ���ʽ��ʾA��C��ɵĻ�������γɹ���Ϊ______________________��
��2��B![]() ���ӵĵ���ʽ��
���ӵĵ���ʽ�� ![]() ����B�����Ӿ�����ͬ�������ķ����У���һ�ַ���
����B�����Ӿ�����ͬ�������ķ����У���һ�ַ���
�������ữ�������Σ��÷��ӵĵ���ʽ�� ![]() ��
��
��3������![]() ������C�������ڵ�ͬ��Ԫ���γɵ���̬�⻯���У��е���ߵ��� ������
������C�������ڵ�ͬ��Ԫ���γɵ���̬�⻯���У��е���ߵ��� ������
�⻯�ﻯѧʽ����ԭ���� ��
��4��д��B����������ˮ�����C����������ˮ����֮�䷴Ӧ�����ӷ���ʽ����
���漰�ĺ�CԪ�ص����ʾ�������ˮ���� ��
(1) MgCl2 ������
(2) Al3+ ![]()
(3) HF HF����֮��������
��4�� Al(OH)3+3H+=Al3++3H2O

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ

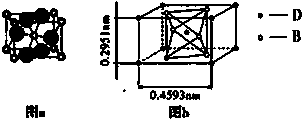
 ��ͼ�����ڱ��ж����ڵ�һ���֣�A��B��C����Ԫ�ص�ԭ�Ӻ��������֮�͵���B����������2��������Ԫ�ط��ű�ʾ��
��ͼ�����ڱ��ж����ڵ�һ���֣�A��B��C����Ԫ�ص�ԭ�Ӻ��������֮�͵���B����������2��������Ԫ�ط��ű�ʾ��