��Ŀ����
����A��B��C��D��E����ǿ����ʣ�������ˮ�пɵ�������������ӣ��������Ӳ��ظ�����

��֪��
��A��B����Һ�ʼ��ԣ�C��D��E��Һ�����ԡ�
��A��Һ��E��Һ��Ӧ�����������г���������A��Һ��C��Һ��Ӧֻ������������������������ͬ����
��D��Һ������������Һ��Ӧ���ܲ���������Cֻ����D��Ӧ����������
�Իش��������⣺
(1)A��B��C��D��E�Ļ�ѧʽ�ֱ�Ϊ_______��________��_______��_______��________��
(2)�����£�Ũ�Ⱦ�Ϊ0.1 mol/L ��A��B��Һ��H2O�ĵ���̶ȵĹ�ϵ��A____(�<����>����=��)B��
(3)��100 mL0.1mol/L E��Һ�У���μ���35 mL 2 mol/L NaOH��Һ�����յõ����������ʵ���Ϊ
____mol��
(1)Na2CO3��Ba(OH)2��HCl��AgNO3��Al2(SO4)3
(2)>
(3)0.01
(2)>
(3)0.01

��ϰ��ϵ�д�
�����Ŀ
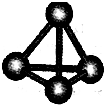 ����A��B��C��D��E��F���ֶ�����Ԫ�أ����ǵ�ԭ��������������D��E���⻯����ӹ��Ͷ���V�ͣ�A��B������������֮����C��������������ȣ�A�ֱܷ���B��C��D�γɵ���������ȵķ��ӣ���A��D���γɵĻ���������¾�ΪҺ̬��
����A��B��C��D��E��F���ֶ�����Ԫ�أ����ǵ�ԭ��������������D��E���⻯����ӹ��Ͷ���V�ͣ�A��B������������֮����C��������������ȣ�A�ֱܷ���B��C��D�γɵ���������ȵķ��ӣ���A��D���γɵĻ���������¾�ΪҺ̬��