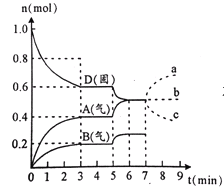
��Ŀ����
����Ŀ��A��B��C��D��E���ֶ�����Ԫ�أ�A��Dͬ���ڣ�A�ĵ��ʼȿ������ᷴӦ���ֿ���NaOH��Һ��Ӧ��B�ĵ����ڷŵ�����������������Ӧ��CԪ�ص����Ӳ������ӣ�DԪ��ԭ�ӵ��������������������������3/4��EԪ��ԭ�ӵ��������������������������3����
��1��A�����ӽṹʾ��ͼΪ_______________��EԪ�������ڱ���λ����_____________________��
��2���õ���ʽ��ʾC����������γɹ���____________________________
��3���Ƚ�B��D��E���⻯��ķе��ɸ����ͣ�_____________________________��
��4�����������Ԫ��A��B��ɣ��������õ��Ե�ԡ������������ˮ������Ӧ���ɻ������ҡ��ҷ����к���10�����ӣ�д���÷�Ӧ�Ļ�ѧ����ʽ��________________�� ��
��5����ѧ�ұ������о��������ǿ��ʱ�������һ�����Թ���:����������еķ��ǻ���ע���ǻ��ǡ�OH����ԭ����������ǿ��������ԡ��躬���ữѧʽΪHnROm,����ǻ���ԭ����Ϊ(m-n)������ֵԽ�ú����������Խǿ��������Ϊ�������ǿ������ǻ���ԭ����(m-n)�����¹�ϵ:
m-n | 0 | 1 | 2 | 3 |
������ǿ�� | �� �� | �� ǿ | ǿ | �� ǿ |
ʵ �� | HClO | H3PO4 | HNO3 | HClO4 |
�ٹ����ж�H2CrO4��HMnO4������ǿ������˳��Ϊ_________________��
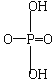
��AsΪ�ڢ�AԪ�أ�H3AsO3Ϊ���ᣬH3PO3Ϊ��ǿ�ᣬ���ƶ�H3AsO3�ķ��ӽṹʽΪ________��H3PO3Ϊ�ĽṹʽΪ__________________��
���𰸡�![]() �ڶ����ڢ�A��
�ڶ����ڢ�A�� ![]() H2O>NH3>H2S AlN + 3H2O��Al(OH)3��+ NH3�� HMnO4��H2Cr2O4
H2O>NH3>H2S AlN + 3H2O��Al(OH)3��+ NH3�� HMnO4��H2Cr2O4 ![]()
��������
A��B��C��D��E���ֶ�����Ԫ�أ�A�ĵ��ʼȿ������ᷴӦ���ֿ���NaOH��Һ��Ӧ����AΪAl��A��Dͬ���ڣ�DԪ��ԭ�ӵ��������������������������![]() ��������������Ϊ6����DΪSԪ�أ�B�ĵ����ڷŵ�����������������Ӧ����BΪNԪ�أ�CԪ�ص����Ӳ������ӣ���CΪHԪ�أ�EԪ��ԭ�ӵ��������������������������3����Eԭ��ֻ����2�����Ӳ㣬����������Ϊ6����EΪOԪ�أ��ݴ˷�����
��������������Ϊ6����DΪSԪ�أ�B�ĵ����ڷŵ�����������������Ӧ����BΪNԪ�أ�CԪ�ص����Ӳ������ӣ���CΪHԪ�أ�EԪ��ԭ�ӵ��������������������������3����Eԭ��ֻ����2�����Ӳ㣬����������Ϊ6����EΪOԪ�أ��ݴ˷�����
A��B��C��D��E���ֶ�����Ԫ�أ�A�ĵ��ʼȿ������ᷴӦ���ֿ���NaOH��Һ��Ӧ����AΪAl��A��Dͬ���ڣ�DԪ��ԭ�ӵ��������������������������![]() ��������������Ϊ6����DΪSԪ�أ�B�ĵ����ڷŵ�����������������Ӧ����BΪNԪ�أ�CԪ�ص����Ӳ������ӣ���CΪHԪ�أ�EԪ��ԭ�ӵ��������������������������3����Eԭ��ֻ����2�����Ӳ㣬����������Ϊ6����EΪOԪ�ء�
��������������Ϊ6����DΪSԪ�أ�B�ĵ����ڷŵ�����������������Ӧ����BΪNԪ�أ�CԪ�ص����Ӳ������ӣ���CΪHԪ�أ�EԪ��ԭ�ӵ��������������������������3����Eԭ��ֻ����2�����Ӳ㣬����������Ϊ6����EΪOԪ�ء�
��1��AΪAl��ԭ�Ӻ�����3�����Ӳ㣬���������Ϊ2��8��3��ʧȥ3�����Ӻ������ӽṹʾ��ͼΪ![]() ��EΪOԪ�أ������ڱ���λ���ǵڶ����ڢ�A�壻
��EΪOԪ�أ������ڱ���λ���ǵڶ����ڢ�A�壻
��2��C��������ΪH2O��Ϊ���ۻ�����õ���ʽ��ʾC����������γɹ���Ϊ![]() ��
��
��3��B��D��E�ֱ�ΪN��S��O���⻯��(Ϊ���Ӿ���)�ķе�һ���ɷ��ӵ���Է���������������Է������е�ߣ���ͬ��Ԫ�ش��ϵ������ԭ����������ģ�����һ����˵��ͬ��Ԫ���⻯��ķе���ϵ���������.�磺�е㣺HCl��HBr��HI������HF�� H2O�� NH3�⼸�����ʵķе��ͬ��Ԫ���⻯��ķе㷴���ظߣ�ԭ������Щ���Ӽ����γ������ͬ�����Ǵ�����Խ��Խ�ߣ���N��S��O���⻯��ķе��ɸ�����ΪH2O>NH3>H2S��
��4�����������Ԫ��Al��N��ɣ��������õ��Ե�ԣ���ΪAlN������ˮ������Ӧ���ɺ�NԪ�صĻ������ң��ҷ����к���10�����ӣ�����ΪNH3����ӦΪˮ�ⷴӦ��ͬʱ���������������÷�Ӧ�Ļ�ѧ����ʽΪ��AlN+3H2O=Al(OH)3��+NH3����
��5����H2CrO4�ķ��ǻ�����2������HMnO4�ķ��ǻ�����3��������H2CrO4Ϊǿ�ᣬHMnO4Ϊ��ǿ�ᣬ������ǿ������˳��ΪHMnO4��H2Cr2O4��
����ΪH3AsO3Ϊ���ᣬ����������в����ڷ��ǻ���������ṹʽӦΪ![]() ��
��
����H3PO3Ϊ��ǿ�ᣬ���ϱ���֪����ǻ�����һ������������ȴ��������ԭ�ӣ�˵������һ����ԭ����ֱ����Pԭ�ӽ�ϣ��÷��ӵĽṹ��ʽΪ ��
��

����Ŀ����ͼ��ʾװ���У��۲쵽������ָ��ƫת��M����֣�N����ϸ���ɴ��жϱ�������M��N��P���ʣ����п��Գ�������

ѡ�� | M | N | P |
A | п | ͭ | ϡ���� |
B | ͭ | �� | ϡ���� |
C | �� | п | ��������Һ |
D | п | �� | ��������Һ |
A. AB. BC. CD. D