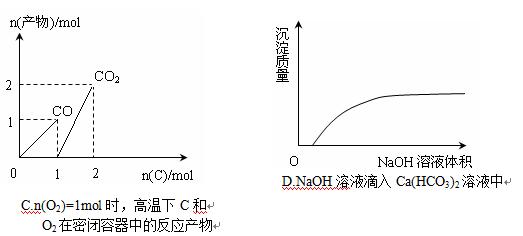
��Ŀ����
(10��)��ҵ�ϳɰ��ķ�Ӧ�� ��һ�����ȷ�Ӧ����֪�÷�Ӧ����2molNH3ʱ���ų�92kJ������.
��һ�����ȷ�Ӧ����֪�÷�Ӧ����2molNH3ʱ���ų�92kJ������.
����ͬ�����£�1 mol N2��3 mol H2�����е����� �������ڡ�����С�ڡ��������ڡ���2 molNH3���е�������
�������1 mol N2��3 mol H2��ϣ�ʹ���ַ�Ӧ���ų������� �������ڡ�����С�ڡ��������ڡ���������ֵ����ԭ���� ��
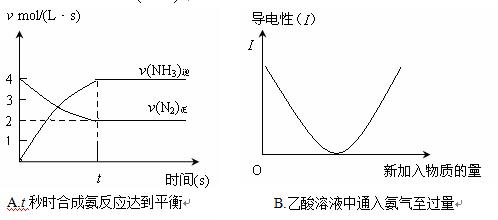
��ʵ����ģ�ҵ�ϳɰ�ʱ��������Ϊ2L���ܱ������ڣ���Ӧ����10min������10 mol NH3������N2��ʾ�ĸ÷�Ӧ����Ϊ�� ��
��һ�������£����ϳɰ��ķ�Ӧ�ﵽ��ѧƽ��ʱ������˵����ȷ����
a.����Ӧ���ʺ��淴Ӧ������� b.����Ӧ��������淴Ӧ����Ϊ0
c.N2��ת���ʴﵽ���ֵ d.N2��H2��Ũ�����
e. N2��H2��NH3������������ f.��Ӧ�ﵽ�����
 ��һ�����ȷ�Ӧ����֪�÷�Ӧ����2molNH3ʱ���ų�92kJ������.
��һ�����ȷ�Ӧ����֪�÷�Ӧ����2molNH3ʱ���ų�92kJ������.����ͬ�����£�1 mol N2��3 mol H2�����е����� �������ڡ�����С�ڡ��������ڡ���2 molNH3���е�������
�������1 mol N2��3 mol H2��ϣ�ʹ���ַ�Ӧ���ų������� �������ڡ�����С�ڡ��������ڡ���������ֵ����ԭ���� ��
��ʵ����ģ�ҵ�ϳɰ�ʱ��������Ϊ2L���ܱ������ڣ���Ӧ����10min������10 mol NH3������N2��ʾ�ĸ÷�Ӧ����Ϊ�� ��
��һ�������£����ϳɰ��ķ�Ӧ�ﵽ��ѧƽ��ʱ������˵����ȷ����
a.����Ӧ���ʺ��淴Ӧ������� b.����Ӧ��������淴Ӧ����Ϊ0
c.N2��ת���ʴﵽ���ֵ d.N2��H2��Ũ�����
e. N2��H2��NH3������������ f.��Ӧ�ﵽ�����

��

��ϰ��ϵ�д�
�����Ŀ
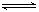 ?mZ(g)����H����a kJ��mol��1(a>0)�����мס������ݻ�����ҹ̶����ܱ��������ڱ��ָ��¶Ⱥ㶨�������£����ܱ���������ͨ��2 mol X��1 mol Y���ﵽƽ��״̬ʱ���ų�����b kJ�����ܱ���������ͨ��1 mol X��0.5 mol Y���ﵽƽ��ʱ���ų�����c kJ����b>2c����a��b��m��ֵ���ϵ��ȷ����(����)
?mZ(g)����H����a kJ��mol��1(a>0)�����мס������ݻ�����ҹ̶����ܱ��������ڱ��ָ��¶Ⱥ㶨�������£����ܱ���������ͨ��2 mol X��1 mol Y���ﵽƽ��״̬ʱ���ų�����b kJ�����ܱ���������ͨ��1 mol X��0.5 mol Y���ﵽƽ��ʱ���ų�����c kJ����b>2c����a��b��m��ֵ���ϵ��ȷ����(����) xC(g)�������� C�ڷ�Ӧ������еİٷֺ�����C%���ͷ�Ӧʱ�䣨t���Ĺ�ϵ��
xC(g)�������� C�ڷ�Ӧ������еİٷֺ�����C%���ͷ�Ӧʱ�䣨t���Ĺ�ϵ��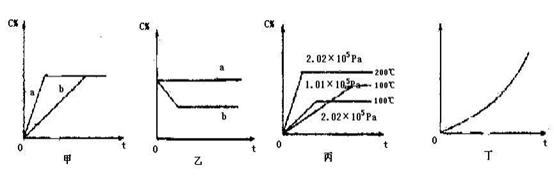
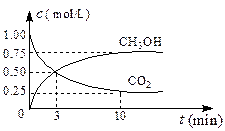
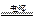 2H2����O2��
2H2����O2��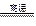 2H2����O2��
2H2����O2�� 2H2����O2��
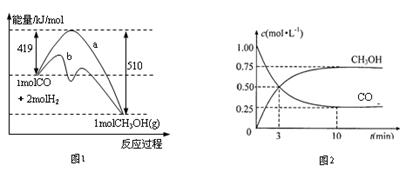
2H2����O2��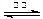 CO��3H2
CO��3H2 CH3OH(g)+H2O(g)
CH3OH(g)+H2O(g) 
 2NH3(g) DH��0��10minʱ�����ʵ�Ũ�Ȳ��ٱ仯�����NH3���ʵ���Ϊ0.4mol��
2NH3(g) DH��0��10minʱ�����ʵ�Ũ�Ȳ��ٱ仯�����NH3���ʵ���Ϊ0.4mol��