��Ŀ����
��16�֣���֪FeSO4�ڲ�ͬ�����·ֽ�õ��IJ��ﲻͬ��������FeO��SO3��Ҳ������Fe2O3��SO3��SO2��ij�о�С��̽���ھƾ���Ƽ���������FeSO4�ֽ����������֪SO3���۵���16.8�棬�е���44.8�档
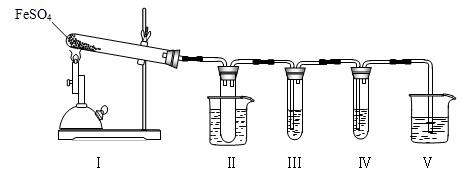
��1��װ�â���Թ��в�װ�κ��Լ�����������______________________
�Թܽ�����50���ˮԡ�У�Ŀ����______________________________
��2��װ�â��װ�â���������̽����ʵ���������ɷ֡������ʵ����ƣ���д�����Լ���Ԥ����������ۡ�
��ѡ�Լ���3 mol��L��1 H2SO4��6 mol��L��1 NaOH��0.5 mol��L��1 BaCl2��0.5 mol��L��1 Ba(NO3)2��
0.01 mol��L��1����KMnO4��Һ��0.01 mol��L��1��ˮ��
��3��װ�â��������Ƿ�ֹβ����Ⱦ�������ձ���Ӧ������Լ��� ��

��1��װ�â���Թ��в�װ�κ��Լ�����������______________________
�Թܽ�����50���ˮԡ�У�Ŀ����______________________________
��2��װ�â��װ�â���������̽����ʵ���������ɷ֡������ʵ����ƣ���д�����Լ���Ԥ����������ۡ�
��ѡ�Լ���3 mol��L��1 H2SO4��6 mol��L��1 NaOH��0.5 mol��L��1 BaCl2��0.5 mol��L��1 Ba(NO3)2��
0.01 mol��L��1����KMnO4��Һ��0.01 mol��L��1��ˮ��
| �����Լ� | Ԥ������ͽ��� |
| װ�â���Թ��м���__________ ___�� | ����������ɫ������֤����������к���SO3�� |
| װ�â����Թ��м���_______ _________�� | ______________________________ ______________________________ ______________________________ ______________________________ |
��16�֣�
��1����ֹ��Һ������װ�â�2�֣� ��ֹSO3Һ�������̣�2�֣�
��2����10�֣�
��
��3��NaOH��Һ��2�֣�
��1����ֹ��Һ������װ�â�2�֣� ��ֹSO3Һ�������̣�2�֣�
��2����10�֣�
| �����Լ� | Ԥ������ͽ��� |
| װ�â���Թ���װ��BaCl2��Һ����3�֣� | ����������ɫ������֤����������к���SO3�� |
| װ�â����Թ���װ������KMnO4��Һ����3�֣� | ����Һ�Ϻ�ɫ��ȥ��֤����������к���SO2����2�֣� ����Һ�Ϻ�ɫ�����Ա仯��֤����������в���SO2����2�֣� |
| �����Լ� | Ԥ������ͽ��� |
| װ�â���Թ���װ��BaCl2��Һ����3�֣� | ����������ɫ������֤����������к���SO3�� |
| װ�â����Թ���װ����ˮ����3�֣� | ����ˮ��ɫ��ȥ��֤����������к���SO2����2�֣� ����ˮ��ɫ�����Ա仯��֤����������в���SO2����2�֣� |
��

��ϰ��ϵ�д�
 ��ս�п�����ϵ�д�
��ս�п�����ϵ�д�
�����Ŀ
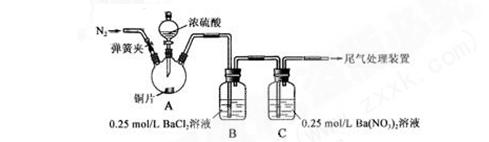
 δ���ְ�ɫ�����ܽ�
δ���ְ�ɫ�����ܽ� Ӧ��
Ӧ�� ��Ϊ֤ʵ���ԵĹ۵㣬��ԭʵ������ϣ�����ԭ�в���֮ǰ����һ���������ò�����_____________;
��Ϊ֤ʵ���ԵĹ۵㣬��ԭʵ������ϣ�����ԭ�в���֮ǰ����һ���������ò�����_____________; ��ϡ���ᡣ������ӷ���ʽ����ʵ��������ͬ��ԭ��_____��
��ϡ���ᡣ������ӷ���ʽ����ʵ��������ͬ��ԭ��_____��
 ���������� ��
���������� ��
 C6H12O6�������ǣ�+6O2��
C6H12O6�������ǣ�+6O2��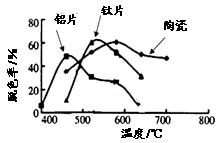

 �����ӵ�±ˮΪ��Ҫԭ���Ʊ���ˮ
�����ӵ�±ˮΪ��Ҫԭ���Ʊ���ˮ ��
�� ���������£�
���������£�
 ��
�� ���ɱ������ݿ�֪�������Ͽ�ѡ��pH���Χ�� ���ữ��ҺZʱ��ʹ�õ��Լ�Ϊ ��
���ɱ������ݿ�֪�������Ͽ�ѡ��pH���Χ�� ���ữ��ҺZʱ��ʹ�õ��Լ�Ϊ ��
 ���壬����װ���к������� ��
���壬����װ���к������� ��
 �ĵ��볣��
�ĵ��볣�� ��
�� ��
�� �ĵ�
�ĵ� �볣��
�볣�� ��
�� ��ijͬѧ���ʵ����֤
��ijͬѧ���ʵ����֤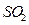 ��
�� ����ǰ�ߵ�
����ǰ�ߵ� ��
�� ��
�� ��
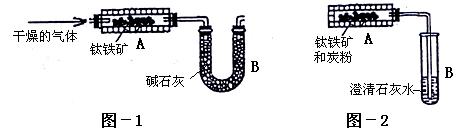
�� ������ˮ������ʯ��ˮ������
������ˮ������ʯ��ˮ������ .
.