��Ŀ����
18������笠ƾ���[5Ca��NO3��2��NH4NO3.10H2O]��������ˮ����һ����ɫ�ĸ��Ϸ��ϣ���1������笠ƾ������Һ�ʼ��ԣ�ԭ����NH4++H2O?NH3•H2O+H+�������ӷ���ʽ��ʾ��
��2����ҵ����������笠Ƶķ������������ȡ��ʯ�õ��Ĵ�����ƣ�������ơ�����Ƽ���泥�Ϊԭ�����Ʊ�������������Ϊͼ1
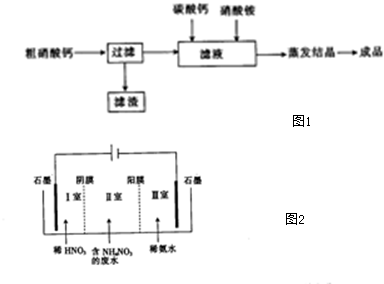
����������Ҫ�ɷ���Ca3��PO4��2���ѧʽ��
�ڼ���������̼��ƺ�����Ӧ�����ӷ���ʽΪCaCO3+2H+�TCa2++CO2��+H2O
���������һ���ֽ�����ʣ����������ʱ��������Ӱ��ϵ��¶ȣ�230��400��ʱ������ֽ����ɿ����к��������������壬�䷴Ӧ�Ļ�ѧ����ʽΪ2NH4NO3$\frac{\underline{\;230��400��\;}}{\;}$2N2��+O2��+4H2O
��3����������笠ƹ����ķ�ˮ�г���������泥�Ŀǰ����ԭ��ⷨ����������ԭ����ͼ2��ʾ����Ĥ����Ĥ�ֱ�ֻ���������ӡ�������ͨ������
�������ĵ缫��ӦʽΪ2H2O-4e-�T4H++O2����I�ҿɻ��յIJ�ƷΪ���ᣨ�����ƣ�
�ڵ������Т�������Һ��pH������������١����䡱��
���� ��1������笠�Ϊ���Σ������Ϊǿ�������Σ�笠�����ˮ�������ԣ�
��2��������ƣ�������ơ������P���ᣩ��ˮ�ܽ⣬���˵ú�����ƺ��������Һ������̼��������ᷴӦ��������ơ�ˮ�Ͷ�����̼���壬��ͨ�백���к�������������泥��������õ���ҺΪ����ƺ�����淋Ļ����Һ�������ᾧ�õ�����笠�[5Ca��NO3��2•NH4NO3•10H2O]���壻
��3���ٸ���Ĥ��ⷨ������Ϊˮʧ���ӷ�����������Ӧ��������������������ʹ������ǿ�����������ƶ����������ϳ����
�ڵ��ʱ������Ϊ����ӦΪ�����ӵõ��ӷ�����ԭ��Ӧ������������ʹ������ǿ�����������ƶ���笠����ӷ�Ӧ���ɰ�����������笠�����Ũ�ȼ�С���ݴ˷�����
��� �⣺��1������笠�Ϊ���Σ�笠�����ˮ�������ԣ���ˮ�����ӷ�ӦΪNH4++H2O?NH3•H2O+H+���ʴ�Ϊ��NH4++H2O?NH3•H2O+H+��
��2���������Ϊ������ˮ�����ʣ�����������Ҫ�ɷ�������ƣ���ѧʽΪCa3��PO4��2���ʴ�Ϊ��Ca3��PO4��2��
�ڼ���̼��������ᷴӦ��������ơ�ˮ�Ͷ�����̼���壬������Ӧ�����ӷ���ʽΪCaCO3+2H+�TCa2++CO2��+H2O��
�ʴ�Ϊ��CaCO3+2H+�TCa2++CO2��+H2O��
�ۿ����к���������������ֱ��ǵ���������������������ԭ��Ӧԭ���ó�����立ֽⷽ��ʽΪ��2NH4NO3$\frac{\underline{\;230��400��\;}}{\;}$2N2��+O2��+4H2O��
�ʴ�Ϊ��2NH4NO3$\frac{\underline{\;230��400��\;}}{\;}$2N2��+O2��+4H2O��
��3����Ĥ��ⷨ������Ϊˮʧ���ӷ�����������Ӧ����ӦʽΪ2H2O-4e-�T4H++O2���������������������������ʹ������ǿ�����������ƶ����������ϳ����ᣬ���Ԣ��ҿɻ��յIJ�ƷΪ���
�ʴ�Ϊ��2H2O-4e-�T4H++O2�������
�ڵ��ʱ������Ϊ����ӦΪ�����ӵõ��ӷ�����ԭ��Ӧ��������������ʹ������ǿ�����������ƶ���笠����ӷ�Ӧ���ɰ�����笠�����ˮ�������ԣ�������笠�����Ũ�ȼ�С�����Ԣ���PH����
�ʴ�Ϊ������
���� ������Ҫ�����˹�ҵ��������笠Ƶ�ԭ������ˮ�Ĵ��������ڿ��鿼���Ķ�����Ϣ����������������������װ�õķ�������缫��Ӧʽ����д���״��㣬��Ŀ�Ѷ��еȣ�

��1��ij�¶��£�Ksp��FeS��=8.1��10-17��FeS������Һ��c��H+����c��S2-��֮����ڹ�ϵ��
c2��H+��•c��S2-��=1.0��10-22��Ϊ��ʹ��Һ��c��Fe2+���ﵽ1mol•L-1���ֽ�����FeSͶ���䱥����Һ�У�Ӧ������Һ�е�pHԼΪB������ĸ����
A��2 B��3 C��4 D��5
��2�������������ϸ�ijЩ���ϻ�ϣ������Ƴɷ������ᣮ�Ի�����Ϊԭ��������������������к�Fe2O3��SiO2��Al2O3��MgO�ȣ����������Ʊ����죨Fe2O3���Ĺ������£�

�����ܹ�����Fe2O3������Ӧ�����ӷ���ʽΪFe2O3+3H2SO4�TFe2��SO4��3+3H2O��������A����Ҫ�ɷֵĻ�ѧʽΪSiO2��
�ڻ�ԭ�����м���FeS2��Ŀ���ǽ���Һ�е�Fe3+��ԭΪFe2+��������������ΪH2SO4������ɸ÷�Ӧ�����ӷ���ʽ��FeS2+14Fe3++8H2O�T15Fe2++2SO42-+16H+
�����������У�O2��NaOH��Fe2+��Ӧ�����ӷ���ʽΪ4Fe2++O2+2H2O+8OH-=4Fe��OH��3����
��Ϊ��ȷ������������ʹ��ȣ�����������Ҫ������Һ��pH�ķ�Χ��3.2��3.8��
| ������ | Fe��OH��3 | Al��OH��3 | Fe��OH��2 | Mg��OH��2 |
| ��ʼ����pH | 2.7 | 3.8 | 7.6 | 9.4 |
| ��ȫ����pH | 3.2 | 5.2 | 9.7 | 12.4 |
����ҺB���Ի��յ�������ABD������ţ���
A��Na2SO4 B��Al2��SO4��3 C��Na2SiO3 D��MgSO4��
| A�� | ${\;}_{6}^{12}C$��${\;}_{6}^{13}C$ | B�� | CH3CH2CH2CH3��CH3CH��CH3��2 | ||
| C�� | ���Ͱ��� | D�� | ���ɱ� |
| A�� | ��֯Ʒ | B�� | ����֯Ʒ | C�� | ë֯Ʒ | D�� | ����֯Ʒ |
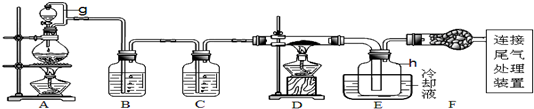
 ijС���Թ�ҵ�����������ᡢ���ᡢ�����Ϊԭ�ϣ��ϳɾ���X[��ѧʽΪFe2��C2O4��3•5H2O]����ȷ���䴿�ȣ�
ijС���Թ�ҵ�����������ᡢ���ᡢ�����Ϊԭ�ϣ��ϳɾ���X[��ѧʽΪFe2��C2O4��3•5H2O]����ȷ���䴿�ȣ�