题目内容
(4分)某同学为探究碳酸钠的性质,进行了如下实验,请你按要求回答有关问题。
(1)配制100 mL 0.5 mol/L的Na2CO3溶液。该同学先用天平称量Na2CO3固体的质量,然后配制该溶液,要用到的玻璃仪器有量筒、烧杯、 、 。
(2)用pH试纸测所配溶液的pH,pH>7,说明Na2CO3溶液呈 (填“酸性”、“碱性”或“中性”);用试管取少量所配溶液,向溶液中滴加几滴酚酞试液,溶液变红,再用酒精灯加热,溶液的颜色变深,说明升高温度,对Na2CO3的水解有 作用。
(1)配制100 mL 0.5 mol/L的Na2CO3溶液。该同学先用天平称量Na2CO3固体的质量,然后配制该溶液,要用到的玻璃仪器有量筒、烧杯、 、 。
(2)用pH试纸测所配溶液的pH,pH>7,说明Na2CO3溶液呈 (填“酸性”、“碱性”或“中性”);用试管取少量所配溶液,向溶液中滴加几滴酚酞试液,溶液变红,再用酒精灯加热,溶液的颜色变深,说明升高温度,对Na2CO3的水解有 作用。
(1)容量瓶、玻璃棒 (2)碱性、促进作用
略

练习册系列答案
相关题目
 实验室有以下的仪器和药品可供使用:
实验室有以下的仪器和药品可供使用: 请回答:
请回答:




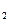 )进行如下实验探究。
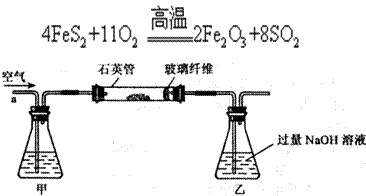
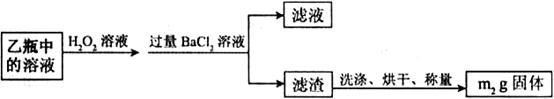
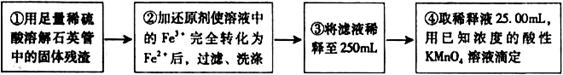
)进行如下实验探究。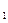 g该黄铁矿样品(杂质中不含硫和铁)放入如下图所示装置(夹持和加热装置省略)的石英管中,从a处不断地缓缓通人空气,高温灼烧石英管中的黄铁矿样品至反应完全。石英管中发生反应的化学方程式为:
g该黄铁矿样品(杂质中不含硫和铁)放入如下图所示装置(夹持和加热装置省略)的石英管中,从a处不断地缓缓通人空气,高温灼烧石英管中的黄铁矿样品至反应完全。石英管中发生反应的化学方程式为: