��Ŀ����
 ���ݻ��̶�Ϊ2L���ܱ������У�����0.18molHI��480��ʱ��Ӧ��
���ݻ��̶�Ϊ2L���ܱ������У�����0.18molHI��480��ʱ��Ӧ��2HI��g��?H2��g��+I2��g������I2����Ϊ��ɫ����ϵ��n��HI����ʱ��仯�����
��
| t/min | 0 | 2 | 4 | 6 | 8 | 10 |
| n��HI��/mol | 0.180 | 0.164 | 0.152 | 0.144 | 0.140 | 0.140 |
��1����ƽ��ʱ��HI��ת������
��2��480��ʱ����Ӧ
| 1 |
| 2 |
| 1 |
| 2 |
��3��Ҫ����Ӧ2HI��g��?H2��g��+I2��g����ƽ�ⳣ�����ɲ�ȡ�Ĵ�ʩ��
A������HI��ʼŨ�� B������������ͨ��I2
C��ʹ�ø�Ч���� D�������¶�
��4����ͼ��480��ʱ������HIŨ����ʱ��ı仯ͼ�����ڸ�ͼ�л����÷�Ӧ��448�淴ӦʱHIŨ�ȵı仯���ߣ�
���㣺��ѧƽ��ļ���,��ѧƽ���Ӱ������
ר�⣺��ѧƽ��ר��
��������������ʽ�����ƽ��ʱ�����ʵ�Ũ�ȣ������������ת���ʺ�ƽ�ⳣ��������Ӱ��ƽ�ⳣ���������жϲ�ȡ�Ĵ�ʩ�������¶ȶ�ƽ���Ӱ��ɻ����仯���ߣ�
���
�⣺��������ʽ��
������������2HI��g�� ?H2��g��+I2��g��
?H2��g��+I2��g��
��ʼŨ�� 0.09mol/L 0 0
ת��Ũ�� 0.02mol/L �� 0.01mol/L 0.01mol/L
ƽ��Ũ�� 0.07mol/L ����0.01mol/L 0.01mol/L
K��=
=
��1��HI��ת����Ϊ
��100%=22.2%���ʴ�Ϊ��22.2%��
��2����Ӧ
H2��g��+
I2��g�� ?HI��g����ƽ�ⳣ��K=
?HI��g����ƽ�ⳣ��K=
=7���ʴ�Ϊ��7��
��3����Ӧ������10min��Ӧ�������¶Ƚ��ͣ�˵���÷�ӦΪ���ȷ�Ӧ������Ӱ��ƽ�ⳣ�������ؿ�֪��ƽ�ⳣ��ֻ���¶��йأ�����Ҫ����ƽ�ⳣ����ֻ�ܸı��¶ȣ���ѡD��
��4����448�淴Ӧʱ��Ӧ����Ҫ��480��ʱС��ƽ�Ҫ�����ƶ�����HI��Ũ��Ҫ��ݴ˿ɻ���ͼΪ��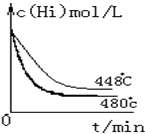 ��
��
�ʴ�Ϊ�� ��
��
������������2HI��g��
 ?H2��g��+I2��g��
?H2��g��+I2��g����ʼŨ�� 0.09mol/L 0 0
ת��Ũ�� 0.02mol/L �� 0.01mol/L 0.01mol/L
ƽ��Ũ�� 0.07mol/L ����0.01mol/L 0.01mol/L
K��=
| 0.01��0.01 |
| 0.072 |
| 1 |
| 49 |
��1��HI��ת����Ϊ
| 0.02 |
| 0.09 |
��2����Ӧ
| 1 |
| 2 |
| 1 |
| 2 |
 ?HI��g����ƽ�ⳣ��K=
?HI��g����ƽ�ⳣ��K=
|
��3����Ӧ������10min��Ӧ�������¶Ƚ��ͣ�˵���÷�ӦΪ���ȷ�Ӧ������Ӱ��ƽ�ⳣ�������ؿ�֪��ƽ�ⳣ��ֻ���¶��йأ�����Ҫ����ƽ�ⳣ����ֻ�ܸı��¶ȣ���ѡD��
��4����448�淴Ӧʱ��Ӧ����Ҫ��480��ʱС��ƽ�Ҫ�����ƶ�����HI��Ũ��Ҫ��ݴ˿ɻ���ͼΪ��
 ��
���ʴ�Ϊ��
 ��
��
������������Ҫ���黯ѧƽ�ⳣ���ļ����Ӧ�ã��ѶȲ�����ʱҪע������ƽ�ⳣ�������弰Ӱ�����أ�

��ϰ��ϵ�д�
�����Ŀ
��������ȿ�����Ũ�������ֿ����ù����ʯ�Ҹ�����ǣ�������
| A��SO2 |
| B��NH3 |
| C��Cl2 |
| D��H2 |
��������������Һ���ȣ�������ų����в�����ϡ����İ�ɫ�������ɵ��ǣ�������
| A���������Һ |
| B���������Һ |
| C���������Һ |
| D����������Һ |
������Һ����Ũ�ȹ�ϵһ����ȷ���ǣ�������
| A��ij�¶��²��CH3COONa��CH3COOH�����ҺpH=7����c��Na+��=c��CH3COO-�� |
| B��0.1mol/L NH4HS��Һ��c��NH4+����c��HS-��+c��H2S��+c��S2-�� |
| C����Ũ�ȵ������NH4Cl��HCl�Ļ����Һ�У�c��Cl-����c��NH4+����c��H+����c��OH-�� |
| D����ͬ�����£�Ũ����ͬ��Na2CO3��C6H5ONa��CH3COONa��NaOH��Һ����pH��ϵΪ��pH ��Na2CO3����pH ��CH3COONa����pH ��C6H5ONa����pH ��NaOH�� |
�е¹��������о������˹��ϳɵĵ�112��Ԫ�ص���ʽ����Ϊ��Copernicium������Ӧ��Ԫ�ط���λ��Cn������Ԫ�ص�������Ϊ�˼�������ѧ�Ҹ������������������˹��ϳ�Cn�Ĺ��̿ɱ�ʾΪ��
Zn+
Pb�T
Cn+
n������������ȷ���ǣ�������
70 30 |
208 82 |
277 112 |
1 0 |
| A�������ϳɹ��������ڻ�ѧ�仯 | ||
| B��CnԪ�ص����ԭ������Ϊ277 | ||
C��
| ||
| D��CnԪ��λ��Ԫ�����ڱ��ĵ������ڣ��Ǹ���Ԫ�� |
�������ӷ���ʽ����д��ȷ���ǣ�������
| A������ϡ���ᷴӦ��2Fe+6H+=2Fe3++3H2�� |
| B���ƺ�ˮ��Ӧ��Na+2H2O=Na++2OH-+H2�� |
| C������FeCl3��Һ��Ӧ��Fe+Fe3+�T2Fe2+ |
| D����������Һ�백ˮ��Ӧ��Al3++3NH3?H2O=Al��OH��3��+3NH4+ |