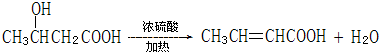��Ŀ����
��I������ѡ����
CH3����CH3����CH3��������Ҫ���л���Ӧ�м��壬�й����ǵ�˵����ȷ���� ��
A�����Ǿ��ɼ���ȥ��һ����ԭ������
B�����ǻ�Ϊ�ȵ����壬̼ԭ�Ӿ���ȡsp2�ӻ�
C��CH3����NH3��H3O+��Ϊ�ȵ����壬���ι��;�Ϊ������
D��CH3���е�̼ԭ�Ӳ�ȡsp2�ӻ�������ԭ�Ӿ�����
E������CH3����һ��CH3����CH3����Ͼ��ɵõ�CH3CH3
��II��
п��һ����Ҫ�Ľ�����п���仯�������Ź㷺��Ӧ�á�
��1��ָ��п�����ڱ��е�λ�ã� ���ڣ� �壬 ����
��2����������п[CH2OH��CHOH��4COO]2Zn��Ŀǰ�г������еIJ�п����д��Zn2+��̬�����Ų�ʽ �������Ƿ�����̼ԭ���ӻ���ʽ�� ��
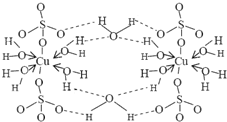
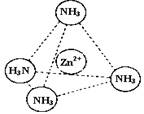
��3��Zn2+����NH3�γ�������[Zn��NH3��4]2+����λ��NH3�������� ������Է��ӡ��Ǽ��Է��ӡ�������[Zn��NH3��4]2+�У�Zn2+λ�������������ģ�Nλ����������Ķ��㣬��������ͼ�б�ʾ[Zn��NH3��4]2+��Zn2+��N֮��Ļ�ѧ����

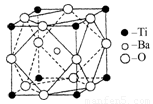
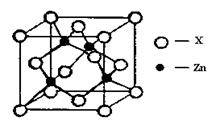
��4������ͼ��ʾп��ij�ǽ���Ԫ��X�γɵĻ����ᄃ��������Zn��Xͨ�����ۼ���ϣ��û�����Ļ�ѧʽΪ ���û�����ľ����۵�ȸɱ��ߵö࣬ԭ���� ��
 ��������ܸ�ϰϵ�д�
��������ܸ�ϰϵ�д�

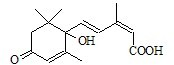 ��I������ѡ����
��I������ѡ����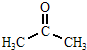 +CH3COOH
+CH3COOH



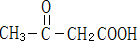
 ��ϵͳ����������
��ϵͳ����������