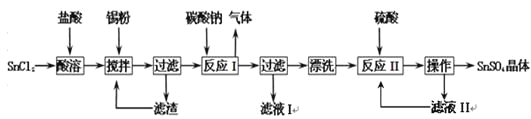
��Ŀ����
����Ŀ����֪A��B��C��D��E�������ڱ���ǰ�����ڵ�Ԫ�أ����ǵĺ˵����A<B<C<D<E������A��B��C��ͬһ���ڵķǽ���Ԫ�ء�������DCΪ���ӻ����D�Ķ�����������C�������Ӿ�����ͬ�ĵ��Ӳ�ṹ��AC![]() Ϊ�Ǽ��Է��ӡ�B��C���⻯��ķе������ͬ����������Ԫ���⻯��ķе�ߡ�E��ԭ�Ӻ�������Ų�δ�ɶԵ���������Ԫ�أ�ECl
Ϊ�Ǽ��Է��ӡ�B��C���⻯��ķе������ͬ����������Ԫ���⻯��ķе�ߡ�E��ԭ�Ӻ�������Ų�δ�ɶԵ���������Ԫ�أ�ECl![]() ����B��C���⻯���γ�����λ��������B��C���⻯�����ʵ���֮��Ϊ2��1������������λ����硣���������������ش��������⣺������ʱ��A��B��C��D��E������Ӧ��Ԫ�ط��ű�ʾ��
����B��C���⻯���γ�����λ��������B��C���⻯�����ʵ���֮��Ϊ2��1������������λ����硣���������������ش��������⣺������ʱ��A��B��C��D��E������Ӧ��Ԫ�ط��ű�ʾ��
��1��A��B��C�ĵ縺����С�����˳��Ϊ ��
��2��A���⻯����һ�����ΪA2H2����,�˷���A��ԭ�ӹ�����ӻ�����Ϊ ��1 mol A2H2���Ҽ�����ĿΪ ��
��3��д��������AC2�ĵ���ʽ ��һ����B��C��ɵĻ�������AC2��Ϊ�ȵ����壬�仯ѧʽΪ ��
��4��E�ļ۵����Ų�ʽ�� ��ECl3��B��C���⻯���γɵ������Ļ�ѧʽΪ
��5��B������������Ӧ��ˮ�����ϡ��Һ��D�ĵ��ʷ�Ӧʱ��B����ԭ����ͼۣ��÷�Ӧ�Ļ�ѧ����ʽ�� ��
���𰸡���1��C<N<O ��2����
��2��sp�ӻ���2���� 3NA ��3��6.02��1023����1����
��3��![]() ��2���� N2O ��1����
��2���� N2O ��1����
��4��3d54S1��2���� ��Cr��NH3��4��H2O��2��Cl3 ��2����
��5��4Mg+10HNO3=4 Mg��NO3��2 +NH4NO3 +3H2O��2����
��������
���������A��B��C��D��E�������ڱ���ǰ�����ڵ�Ԫ�أ����ǵĺ˵����A��B��C��D��E��������DC�ľ���Ϊ���Ӿ��壬D�Ķ�����������C�������Ӿ�����ͬ�ĵ��Ӳ�ṹ��C�γ�-2�������ӣ���Dλ��C����һ���ڣ�B��C���⻯��ķе������ͬ����������Ԫ���⻯��ķе�ߣ�������Ӧ���������C�γ�-2�������ӣ���CΪ��Ԫ�أ�DΪþԪ�أ��˵����B��C����BΪ��Ԫ�أ�����A��B��C��ͬһ���ڵķǽ���Ԫ�أ�AC2Ϊ�Ǽ��Է��ӣ���AΪ̼Ԫ�أ�E��ԭ�Ӻ�������Ų�δ�ɶԵ���������Ԫ�أ���E��ԭ������Ϊ24��EΪCrԪ�أ�CrCl3����NH3��H2O�γ�����λ��������������������ʵ���֮��Ϊ2��1������������4��NH3��2��H2O������������λ����磬�������Ϊ[Cr��NH3��4��H2O��2]Cl3��
��AΪ̼Ԫ�أ�BΪ��Ԫ�أ�CΪ��Ԫ�أ�DΪþԪ�أ�EΪCrԪ�ء�
��1��AΪ̼Ԫ�ء�BΪ��Ԫ�ء�CΪ��Ԫ�أ�ͬ����������ҵ縺����������縺����С�����˳��ΪC<N<O��
��2��A���⻯����һ�����ΪA2H2������C2H2���˷�����Cԭ���γ�2���Ҽ���û�й¶Ե��ӣ�����C��ԭ�ӹ�����ӻ�����Ϊsp�ӻ�����Ȳ�ĽṹʽΪH-C��C-H��1����Ȳ��������3���Ҽ�����1 molCH��CH�����к��Ҽ�����ĿΪ3NA.
��3��������AC2��CO2��������̼ԭ������ԭ��֮���γ�2�Թ��õ��Ӷԣ�����ʽΪ![]() ��һ����NԪ�ء�OԪ�ػ�������CO2��Ϊ�ȵ����壬�仯ѧʽΪN2O��
��һ����NԪ�ء�OԪ�ػ�������CO2��Ϊ�ȵ����壬�仯ѧʽΪN2O��
��4��EΪCrԪ�أ�ԭ������Ϊ24��ԭ�Ӻ�����24�����ӣ��۵����Ų�ʽ��3d54S1��CrCl3����NH3��H2O�γ�����λ��������������������ʵ���֮��Ϊ2��1������������4��NH3��2��H2O������������λ����磬�������Ϊ[Cr��NH3��4��H2O��2]Cl3��
��5�� B������������Ӧ��ˮ�����ϡ��ҺΪHNO3��D�ĵ���ΪMg��������Mg��Ӧʱ��N����ԭ����ͼۣ�����������泥����ݵ�ʧ�����غ��ԭ���غ���ƽ���÷�Ӧ����ѧ����ʽ��4Mg+10HNO3=4 Mg��NO3��2 +NH4NO3 +3H2O��

 ��������ϵ�д�
��������ϵ�д�