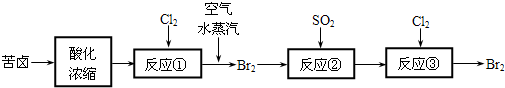
��Ŀ����
�ۻ�ѧ��ѡ��ѧ�뼼���ݣ�15�֣���ش��й��ȼҵ���������⣺
��1���������ռ��ǵ��ʳ��ˮʱ���չ̶��ı���k�������ȣ����ɵIJ�Ʒ��������k���ߣߣߣߣ�Ҫ��������ʽ�ͽ����;
��2��ԭ�ϴ����г�������ɳ��Ca2����Mg2����Fe3����SO42�������ʣ����뾫�ƺ���ܹ����ʹ�á�����ʱ����������ˮ���˺�Ҫ������Լ��ֱ�Ϊ��Na2CO3����HCl�����ᣩ��BaCl2����3���Լ����ӵĺ���˳���ǣߣߣߣߣߣߣߣߣ�����ţ�
��3���ȼҵ�Ǹߺ��ܲ�ҵ��һ�ֽ�������ȼ�ϵ������ϵ��¹��տ��Խڣ��磩��30�����ϡ������ֹ�������У�������ϵĴ�����ת����ϵ����ͼ��ʾ�����еĵ缫δ��������õ�����Ĥ��ֻ����������ͨ����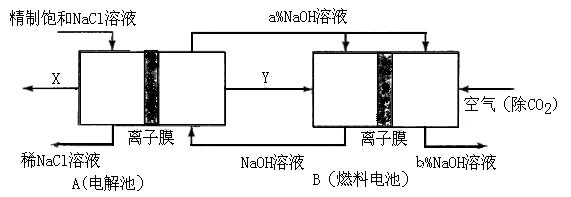
��ͼ��X��Y�ֱ��ǣߣߣߣߡ��ߣߣߣߣ��ѧʽ���������Ƚ�ͼʾ������������������a����b���Ĵ�С_�ߣߣߣߣ�
�ڷֱ�д��ȼ�ϵ��B�������������Ϸ����ĵ缫��Ӧ�������ߣߣߣߣ��������ߣߣߣߣ���������Ƶ���Ҫ�ڣ��磩��֮�����ڣ�д��2�����ߣߣߣߣߣߣߣߡ��ߣߣߣߣߣߣߣߡ�
��1��k=M��Cl2��/2 M��NaOH��=71/80=1:1��13��0��89��2�֣�
��2���ۢ٢ڣ�2�֣���3����Cl2 H2 a��С��b�� ��3�֣���O2+4e-+2H2O��4OH- 2H2��4e-+4OH-��2H2O ��4�֣���ȼ�ϵ�ؿ��Բ���������ĵĵ��ܣ�2�֣�����߲�����Һ��Ũ�ȣ������ܺģ�����������Ҳ���֣���2�֣�
����

��ͼ��ij�����Ժ�ˮ��Դ�����ۺ����õ�ʾ��ͼ��
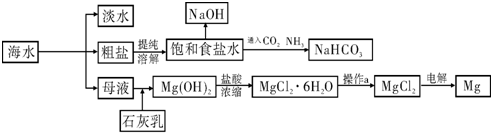
��ش��������⣺
��1�����о�һ�ֺ�ˮ�����ķ���
��2����ҵ�ϳ������ӽ���Ĥ��������NaOH���������д���ͨ�����ӽ���Ĥ��������
��3�������Ƽ��������ʳ��ˮ��ͨ��CO2��NH3�Ƶ�NaHCO3����ͨ��
���ѧʽ����������
��4��þ��һ����;�ܹ㷺�Ľ������ϣ�Ŀǰ������60%��þ�Ӻ�ˮ����ȡ����֪�й����ʵ��۷е��������£�
| MgO | MgCl2 | |
| �۵�/�� | 2852 | 714 |
| �е�/�� | 3600 | 1412 |
 2NH3��+CaCl2+2H2O
2NH3��+CaCl2+2H2O Na2CO3+CO2��+H2O
Na2CO3+CO2��+H2O