��Ŀ����
X��Y��Z��L��M����Ԫ�ص�ԭ��������������X��Y��Z��L����ɵ����ʵĻ���Ԫ�أ�M�ǵؿ��к�����ߵĽ���Ԫ�ء�
�ش��������⣺
(1)L��Ԫ�ط���Ϊ________��M��Ԫ�����ڱ��е�λ��Ϊ________������Ԫ�ص�ԭ�Ӱ뾶�Ӵ�С��˳����________(��Ԫ�ط��ű�ʾ)��
(2)Z��X��Ԫ�ذ�ԭ����Ŀ��1��3��2��4���ɷ���A��B��A�ĵ���ʽΪ________��B�ĽṹʽΪ________��������������������������������������
(3)��(Se)������������Ԫ�أ���Lͬһ���壬Seԭ�ӱ�Lԭ�Ӷ��������Ӳ㣬��Se��ԭ������Ϊ________��������������Ӧ��ˮ���ﻯѧʽΪ________���������������Ԫ�ص��ʷֱ���H2��Ӧ����1 mol��̬�⻯��ķ�Ӧ�����£���ʾ����1 mol�����ⷴӦ�ȵ���________(����ĸ����)��
a����99.7 kJ��mol��1 b����29.7 kJ��mol��1 c����20.6 kJ��mol��1 d����241.8 kJ��mol��1
(4)��M������������ʯī��������NaHCO3��Һ�����Һ���е�⣬����������R��R���ȷֽ����ɻ�����Q��д����������R�ĵ缫��Ӧʽ��____________________________________����R����Q�Ļ�ѧ����ʽΪ________________________________________________________��
��(1)O���������ڢ�A�塡Al>C>N>O>H
(2) 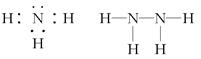 ��(3)34��H2SeO4��b
��(3)34��H2SeO4��b
(4)Al��3HCO3����3e��===Al(OH)3����3CO2��[��Al��3e��===Al3����Al3����3HCO3��===Al(OH)3����3CO2��]��2Al(OH)3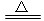 Al2O3��3H2O
Al2O3��3H2O
����

X��Y��Z��W��R��Ԫ�����ڱ�ǰ�������еij���Ԫ��,�������Ϣ���±�:
| Ԫ�� | �����Ϣ |
| X | ��ɵ����ʵĻ���Ԫ��,����������ϼ���������ϼ۵Ĵ�����Ϊ2 |
| Y | �ؿ��к�����ߵ�Ԫ�� |
| Z | ����������Ϊ23,������Ϊ11�ĺ��� |
| W | �����д���ʹ����Ͻ���Ʒ,��ҵ�Ͽ��õ������������ķ����Ʊ��䵥�� |
| R | �ж��ֻ��ϼ�,���ɫ���������ڿ����л�Ѹ�ٱ�ɻ���ɫ,����ɺ��ɫ |
(1)W��Ԫ�����ڱ��е�λ��Ϊ������;X��Y��Z��W����Ԫ�ص�ԭ�Ӱ뾶�Ӵ�С��˳������������������(��Ԫ�ط��ű�ʾ)��
(2)X������Ԫ�ذ�ԭ����Ŀ��1��3��2��4���ɷ���A��B,A�ĽṹʽΪ������;B�ĵ���ʽΪ����������������ZY�д��ڵĻ�ѧ������Ϊ����������
(3)��(As)������������Ԫ��,��Xͬһ����,Asԭ�ӱ�Xԭ�Ӷ��������Ӳ�,�����ԭ������Ϊ������,������������Ӧ��ˮ����Ļ�ѧʽΪ���������������������������Ԫ�ص���̬�⻯����ȶ��ԴӴ�С��˳������������������������������(�û�ѧʽ��ʾ)��
(4)��RCl3��Һ��ʴͭ��·������ӷ���ʽΪ����������������������������������Һ��R3+���õ��Լ�����������,���Թ۲쵽����������������������������
(5)Z
 W�Ͻ�(Z17W12)��һ��DZ�ڵ��������,��Z��W������һ���������������ɡ��úϽ���һ����������ȫ����ķ�Ӧ����ʽΪ:Z17W12+17H2
W�Ͻ�(Z17W12)��һ��DZ�ڵ��������,��Z��W������һ���������������ɡ��úϽ���һ����������ȫ����ķ�Ӧ����ʽΪ:Z17W12+17H2 17ZH2+12W,�õ��Ļ����Q(17ZH2+12W)��6.0 mol��L-1 HCl��Һ������ȫ�ͷų�H2��1 mol Z17W12��ȫ�����õ��Ļ����Q������������ȫ��Ӧ,�ͷų�H2�����ʵ���Ϊ��������
17ZH2+12W,�õ��Ļ����Q(17ZH2+12W)��6.0 mol��L-1 HCl��Һ������ȫ�ͷų�H2��1 mol Z17W12��ȫ�����õ��Ļ����Q������������ȫ��Ӧ,�ͷų�H2�����ʵ���Ϊ�������� ������Ԫ��X��Y��Z��M��Nԭ���������������й���Ϣ���£�
| Ԫ�� | �й���Ϣ |
| X | �����������Ǵ�����2�� |
| Y | Ԫ�ص���Ҫ���ϼ�Ϊ-2�� |
| Z | �䵥�ʼ����������ɫΪ��ɫ |
| M | ��Xͬ���壬�䵥��Ϊ�뵼����� |
| N | �䵥����Y������ȼ�գ���������������ɫ���� |
��1��XY2�ĵ���ʽ�� ��Z2Y2��������ѧ�������� ��XY2��Z2Y2��Ӧ�Ļ�ѧ����ʽ�� ��
��2�����������£�һ������NY2��Y2������Ӧ��2NY2��g��+Y2��g��
 2NY3��g�����¶ȷֱ�ΪT1��T2ʱ��NY3�����������ʱ��仯����ͼ���÷�Ӧ�ġ�H 0�����������<����=������ͬ������T1��T2ʱ�÷�Ӧ�Ļ�ѧƽ�ⳣ���ֱ�ΪK1��K2����K1 K2��
2NY3��g�����¶ȷֱ�ΪT1��T2ʱ��NY3�����������ʱ��仯����ͼ���÷�Ӧ�ġ�H 0�����������<����=������ͬ������T1��T2ʱ�÷�Ӧ�Ļ�ѧƽ�ⳣ���ֱ�ΪK1��K2����K1 K2��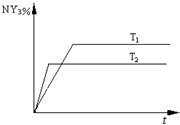
��3��X�ķǽ����Ա�M ���ǿ��������������֤���˽��۵���ʵ�� �������ӷ���ʽ��ʾ����
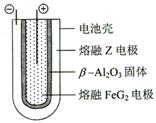
 Fe + 2ZG
Fe + 2ZG 
