��Ŀ����
��12�֣�ij�о���ѧϰС�齫����װ����ͼ���ӣ�C��D��E��F��X��Y ���Ƕ��Ե缫�������е����̪��Һ������Դ��ͨ����F�������Ժ�ɫ���Իش���������:
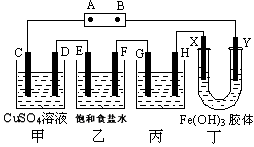
��1����ԴA ����������___________��
��2����װ���е�ⷴӦ�� �ܻ�ѧ����ʽ��
�ܻ�ѧ����ʽ��
______________________________��
��3������ռ���װ���в��������壬����������������_________ _��
_��
��4�����ñ�װ�ø�ͭ������GӦ����______���ͭ���������������Һ��_______���
ѧʽ����Һ��
��5��װ�ö��е�������________________________________________��

��1����ԴA ����������___________��
��2����װ���е�ⷴӦ��
 �ܻ�ѧ����ʽ��
�ܻ�ѧ����ʽ��______________________________��
��3������ռ���װ���в��������壬����������������_________
 _��
_����4�����ñ�װ�ø�ͭ������GӦ����______���ͭ���������������Һ��_______���
ѧʽ����Һ��
��5��װ�ö��е�������________________________________________��
��1������
(2) 2CuSO4+2H2O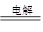 2Cu+O2��+2H2SO4
2Cu+O2��+2H2SO4
��3��1:1
��4���� AgNO3
��5��Y���������ɫ����
(2) 2CuSO4+2H2O
 2Cu+O2��+2H2SO4
2Cu+O2��+2H2SO4 ��3��1:1
��4���� AgNO3
��5��Y���������ɫ����
��

��ϰ��ϵ�д�
 ��ĩ���100�ִ��½����ȫ�Ծ�ϵ�д�
��ĩ���100�ִ��½����ȫ�Ծ�ϵ�д�
�����Ŀ
 ����ȫ���գ��Ƶ��н�ǿɱ������������Һ(��84����Һ��)���������ͼ��װ�ã���Ե�Դ�缫���ƺ�����Һ����Ҫ�ɷ��ж���ȷ����
����ȫ���գ��Ƶ��н�ǿɱ������������Һ(��84����Һ��)���������ͼ��װ�ã���Ե�Դ�缫���ƺ�����Һ����Ҫ�ɷ��ж���ȷ����