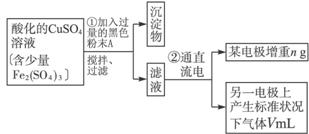
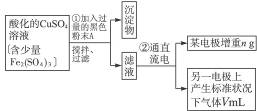
��Ŀ����
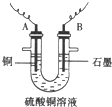
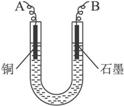
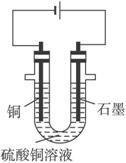
��֪��pHΪ4-5�Ļ����У�Cu2+������ˮ�⣬��Fe3+������ȫˮ�⣮ijѧ�����õ�ⴿ����CuSO4��Һ�ķ��������ݵ缫������ͭ�������Լ��缫�ϲ��������������ⶨͭ�����ԭ����������ʵ���������ͼ��ʾ��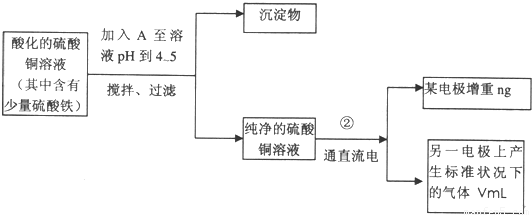
��1������������A������______
a��NH3?H2O b��Cu c��CuO d��Cu��OH��2
��2�����˲������õ��IJ���������______������������õIJ���������ͼ����A����ֱ����Դ��______����B�缫�Ϸ����ĵ缫��ӦΪ______��
��3����ʼһ��ʱ�����U�ι��й۲쵽��������______�������ӷ���ʽΪ______ 2Cu+4H++02��
���𰸡���������1��������ˮ�⣬����pH��ʹ������ת��Ϊ��������ȥ��
��2��������Ҫ©�����ձ�����������������A������Cu����AΪ������BΪ������
��3����ⷢ��2Cu2++2H2 2Cu+4H++02�����Դ˷�������
2Cu+4H++02�����Դ˷�������
��4�����ݵ缫������ͭ�������Լ��缫�ϲ��������������ⶨͭ�����ԭ������������Ҫ�������ǰ��缫��������
��5�����ݵ����غ��֪��2Cu��02�����Դ˼��㣮
����⣺��1��������ˮ�⣬����pH��ʹ������ת��Ϊ��������ȥ��Cu�������Ӳ���Ӧ���Ӱ�ˮ���������ʣ�ѡcd����������ˮ��ƽ�������ƶ�����ȫת��Ϊ������
�ʴ�Ϊ��cd��
��2��������Ҫ©�����ձ�����������������A������Cu����AΪ���������Դ����������BΪ������������ӦΪ40H--4e-�T02��+2H20��
�ʴ�Ϊ���ձ�����������©�������� 40H--4e-�T02��+2H20��
��3����ⷢ��2Cu2++2H2 2Cu+4H++02�����۲쵽ͭ����֣�ʯī�������������ɣ���Һ����ɫ��dz��
2Cu+4H++02�����۲쵽ͭ����֣�ʯī�������������ɣ���Һ����ɫ��dz��
�ʴ�Ϊ��ͭ����֣�ʯī�������������ɣ���Һ����ɫ��dz��2Cu2++2H2 2Cu+4H++02����
2Cu+4H++02����
��4�����ݵ缫������ͭ�������Լ��缫�ϲ��������������ⶨͭ�����ԭ������������Ҫ�������ǰ��缫��������
A��ʵ��֮ǰӦ�������ǰ�缫������������ȷ
B�����缫�ں�ɳ���ǰ������������ˮ��ϴ������������ȷ��
C�����µ���缫��������ͭ������ϴ�����أ���������ȷ���ѵõ�ȷ��Cu���������ʴ���
D���缫�ں�ɳ��صIJ����б��밴�����-����-�ٺ��-�ٳ��ء����У���ֹCu������������ȷ��
E�����п������ڵ�����£���ɵ缫������õ��º�ɵķ�������ֹCu������������ȷ��
�ʴ�Ϊ��ABDE��
��5����Cu�����ԭ������Ϊx��
���ݵ����غ��֪��2Cu��02������
2x 1
ng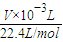
2x×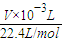 =n��
=n��
���x=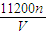 ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
���������⿼��ʵ�鷽������ƣ��漰���ԭ�������ԭ�������IJⶨ��ע�����е����غ㼴�ɽ�𣬣�4��Ϊ�����״��㣬��Ŀ�Ѷ��еȣ�
��2��������Ҫ©�����ձ�����������������A������Cu����AΪ������BΪ������
��3����ⷢ��2Cu2++2H2
 2Cu+4H++02�����Դ˷�������
2Cu+4H++02�����Դ˷���������4�����ݵ缫������ͭ�������Լ��缫�ϲ��������������ⶨͭ�����ԭ������������Ҫ�������ǰ��缫��������
��5�����ݵ����غ��֪��2Cu��02�����Դ˼��㣮
����⣺��1��������ˮ�⣬����pH��ʹ������ת��Ϊ��������ȥ��Cu�������Ӳ���Ӧ���Ӱ�ˮ���������ʣ�ѡcd����������ˮ��ƽ�������ƶ�����ȫת��Ϊ������
�ʴ�Ϊ��cd��
��2��������Ҫ©�����ձ�����������������A������Cu����AΪ���������Դ����������BΪ������������ӦΪ40H--4e-�T02��+2H20��
�ʴ�Ϊ���ձ�����������©�������� 40H--4e-�T02��+2H20��
��3����ⷢ��2Cu2++2H2
 2Cu+4H++02�����۲쵽ͭ����֣�ʯī�������������ɣ���Һ����ɫ��dz��
2Cu+4H++02�����۲쵽ͭ����֣�ʯī�������������ɣ���Һ����ɫ��dz���ʴ�Ϊ��ͭ����֣�ʯī�������������ɣ���Һ����ɫ��dz��2Cu2++2H2
 2Cu+4H++02����
2Cu+4H++02������4�����ݵ缫������ͭ�������Լ��缫�ϲ��������������ⶨͭ�����ԭ������������Ҫ�������ǰ��缫��������
A��ʵ��֮ǰӦ�������ǰ�缫������������ȷ
B�����缫�ں�ɳ���ǰ������������ˮ��ϴ������������ȷ��
C�����µ���缫��������ͭ������ϴ�����أ���������ȷ���ѵõ�ȷ��Cu���������ʴ���
D���缫�ں�ɳ��صIJ����б��밴�����-����-�ٺ��-�ٳ��ء����У���ֹCu������������ȷ��
E�����п������ڵ�����£���ɵ缫������õ��º�ɵķ�������ֹCu������������ȷ��
�ʴ�Ϊ��ABDE��
��5����Cu�����ԭ������Ϊx��
���ݵ����غ��֪��2Cu��02������
2x 1
ng
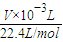
2x×
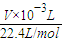 =n��
=n�����x=
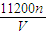 ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
�����������⿼��ʵ�鷽������ƣ��漰���ԭ�������ԭ�������IJⶨ��ע�����е����غ㼴�ɽ�𣬣�4��Ϊ�����״��㣬��Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ