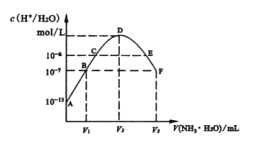
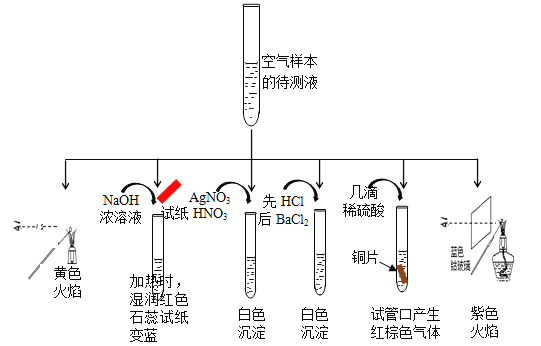
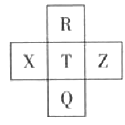
��Ŀ����
����Ŀ������˵�����ʾ������ȷ����( )
A����֪2C(s)+2O2(g)=2CO2(g) ��H=a kJ��mol��1��2C(s)+O2(g)=2CO(g) ��H=b kJ��mol��1����a>b
B����H��(aq)��OH��(aq)=H2O(l) ��H����57.3 kJ��mol��1��֪��������1 mol CH3COOH��ϡ��Һ�뺬1 mol NaOH��ϡ��Һ��ϣ��ų�������С��57.3 kJ
C����C(ʯī)=C(���ʯ) ��H��+1.9 kJ��mol��1��֪�����ʯ��ʯī�ȶ�
D����100 kPaʱ��1 g H2��ȫȼ������Һ̬ˮ���ų�285.8 kJ��������H2ȼ�յ��Ȼ�ѧ����ʽ��ʾΪ��2H2(g)��O2(g)=2H2O(l) ��H����571.6 kJ��mol��1
���𰸡�B
��������
���������A��һ����̼ȼ�ջ�ų��������ų�������Խ����HԽС����a��b����A����B����ϡ��Һ�У�H++OH-�TH2O��H=-57.3 kJmol�������������ᣬ������Ҫ�����������뺬1 mol NaOH����Һ��ϣ��ų�������С��57.3 kJ����B��ȷ��C������C(ʯī)�TC(���ʯ)��H=+1.9kJ/mol����֪ʯī���������ڽ��ʯ������ʯī�Ƚ��ʯ�ȶ�����C����D��101 kPaʱ��1gH2 �����ʵ���Ϊ0.5mol����ȫȼ������Һ̬ˮ���ų�285.8 kJ����������ȼ�յ��Ȼ�ѧ����ʽ��ʾΪ��H2(g)+![]() O2(g)�TH2O(l)��H=-517.6 kJ/mol����D����ѡB��
O2(g)�TH2O(l)��H=-517.6 kJ/mol����D����ѡB��