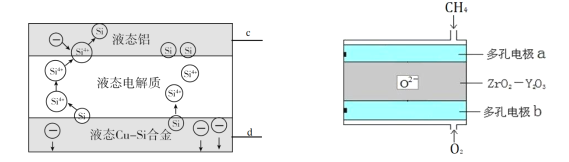
��Ŀ����
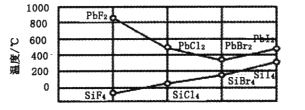
����Ŀ����֪��1mol�����еĻ�ѧ����Ҫ����436kJ��������1mol�����еĻ�ѧ����Ҫ����498kJ����������ͼ�е�����ͼ���ش��������⣺

��1���ֱ�д���٢ڵ���ֵ��
��____________�� �� ____________ ��
��2������H2O��g���е�1mol H-O���ų�____________kJ��������
��3����֪��H2O��l��= H2O��g�� ��H = +44 kJ��mol��1 ����д����������������ȫȼ������Һ̬ˮ���Ȼ�ѧ����ʽ��_____________________________________��
���𰸡�1370 1852 463 2H2��g��+O2(g) == 2H2O��l����H= ��570kJ/mol
��������
��ͼ���֪2H2��g��+O2��g��=2H2O��g����H=-482kJmol-1����ѧ��Ӧ��ʵ���Ǿɼ��Ķ��Ѻ��¼����γɣ���ѧ������Ҫ�����������γ��¼�Ҫ�ų���������Ӧ��Ϊ��Ӧ����ܼ��ܼ�ȥ��������ܼ��ܣ��Դ˽����⡣
��1�����У���֪��1 mol�����еĻ�ѧ����Ҫ����436 kJ��������1 mol�����еĻ�ѧ����Ҫ����498 kJ ���������Բ�2Ħ��������1Ħ����������Ҫ������Ϊ2��436+498=1370kJ�����и��������غ㣬����֪���ų�������Ϊ1370+482=1852kJ��
��2������H2O��g����1 mol H��O���ų�������Ϊ 1852 /4=463kJ��
��3����ͼ����Ϣ����֪����2H2��g��+O2��g��=2H2O��g����H =-482kJ��mol-1��
����ΪH2O��l��=H2O��g����H = +44 kJ��mol-1������2 mol������������������ȫȼ������Һ̬ˮ���Ȼ�ѧ����Ϊ2H2��g��+O2��g��=2H2O��l����H =-570 kJ��mol-1��

 ͬ������ϵ�д�
ͬ������ϵ�д�