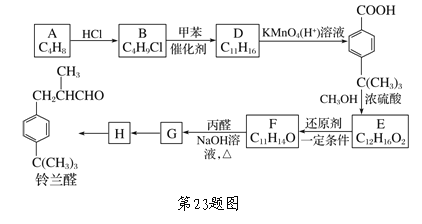
��Ŀ����
����Ŀ�����ݻ�ѧ��Ӧ������ת�������֪ʶ���Իش���������:
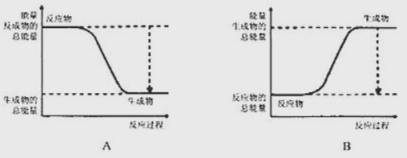
��1����֪![]() �����ױȰ����ȶ����ṹ��ʯī���ƣ���ͼ����ȷ��ʾ�÷����������仯����___������ţ���
�����ױȰ����ȶ����ṹ��ʯī���ƣ���ͼ����ȷ��ʾ�÷����������仯����___������ţ���
��2����֪������ȼ�ϵ�ص��ܷ�Ӧ����ʽΪ2H2O+O2=2H2O���ڼ��������£�ͨ������һ�˵ĵ缫��ӦʽΪ_________________����·��ÿת��0.2mol���ӣ���״��������H2�������______L��
��3����֪�Ͽ�1mol N��N����Ҫ946kJ���������Ͽ�1mol H��H����Ҫ436kJ������������1moN��H���ų�391kJ���������Լ�������2mol NH3ʱ��______����ų��������ա���______kJ������������ͬ���������������г���1molN2��3molH2ʱ�����Ƿ�Ӧ��Ӧ������______������ڡ��������ڡ���С�ڡ��������������ֵ��ԭ����______��
���𰸡���1��A
��2��H2-2e-+2OH- =2H2O ��2.24��
��3���ų���92��С�ڣ����淴Ӧ����Ӧ�ﲻ����ȫת��Ϊ��������
��������
�����������1�����ʺ��е�����Խ�ͣ����ʵ��ȶ��Ծ�Խǿ�����ں��ױȰ����ȶ����������������\�ĸߣ�����ת��Ϊ�\�ķ�Ӧ�Ƿ��ȷ�Ӧ������ȷ��ʾ�÷����������仯����Aͼ��2������ȼ�ϵ�ص��ܷ�Ӧ����ʽΪ2H2O+O2=2H2O���ڼ��������£�ͨ������һ���Ǹ����������ĵ缫��ӦʽΪH2-2e-+2OH- =2H2O�������ܷ�Ӧ����ʽ��֪��ÿ��2mol����������Ӧ��ת�Ƶ��ӵ����ʵ�����4mol�����·��ÿת��0.2mol���ӣ���Ӧ���ı�״����H2�����ʵ�����0.1mol���������V=0.1mol�� 22.4L/mol=2.24L����3����֪�Ͽ�1mol N��N����Ҫ946kJ���������Ͽ�1mol H��H����Ҫ436kJ������������1moN��H���ų�391kJ������������ݷ�Ӧ����ʽN2(g)+3H2(g)=2NH3(g)��֪������2mol NH3ʱH=946kJ/mol+3��436kJ/mol-6��391kJ/mol=-92kJ/mol�������ų�92kkJ������������ͬ���������������г���1molN2��3molH2ʱ�����ڸ÷�Ӧ�ǿ��淴Ӧ����Ӧ�ﲻ����ȫת��Ϊ������������Ƿ�Ӧ��Ӧ������С���������ֵ��

����Ŀ��ʵ�����Ʊ�����ͪ�ķ�Ӧԭ����

�䷴Ӧ��װ��ʾ��ͼ�������г�װ�á�����װ����ȥ����

������������ͪ������ʳ��ˮ��ˮ�IJ����������ʼ��±���ע�������е����ݱ�ʾ���л�����ˮ�γɵľ��й̶���ɵĻ����ķе���
���� | �е������� | �ܶ���g��cm��3, 20���� | �ܽ��� |
������ | 161.1��97.8�� | 0.962 | ������ˮ |
����ͪ | 155.6��95�� | 0.948 | ����ˮ |
����ʳ��ˮ | 108.0 | 1.330 | |
ˮ | 100.0 | 0.998 |
��1��ʵ����ͨ��װ��B������Na2Cr2O7��Һ�ӵ�ʢ�л�������A�У���55��60�����з�Ӧ����Ӧ��ɺ�������ˮ�������ռ�95 ~ 100������֣��õ���Ҫ������ͪ��Ʒ��ˮ�Ļ���
�� ����B�������� ��
�� �������ʱ��һ��ʱ�����δͨ����ˮ��Ӧ��ȡ����ȷ������ ��
�� �����ܷ��뻷��ͪ��ˮ��ԭ���� ��
��2������ͪ���ᴿ��Ҫ��������һϵ�еIJ�����
a�������ռ�151~ 156����֣��õ���Ʒ
b������
c�����ռ����Ĵ�Ʒ�м�NaCl���������ͣ����ã���Һ
d��������ˮMgSO4���壬��ȥ�л���������ˮ
�� ������������ȷ˳���� �����������
�� ����������c������NaCl����������� ��
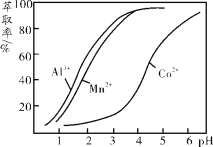
��3������ͪ��һ�ֳ��õ���ȡ�����Ի���ͪΪ�ɷ�֮һ����ȡҺ�Խ�����������ȡ����pH�Ĺ�ϵ����ͼ������ij�ܿ�ʯ���������Һ, ����Һ���е���������Ҫ��H+��Co2+��Mn2+��Al3+����ȥAl3+ ����������ȡ�������MnCl2�Եõ���Ϊ������CoCl2��Һ��pH��Χ������ ��
a��2.0��2.5 b��3.0��3.5 c��4.0��4.5
��4����ʵ����ԭ��������20mL������������Na2Cr2O7��Һ10mL�����ճƵò�Ʒ����Ϊ14.7g������������ͪ�IJ���Ϊ ���ٷ�����ȷ��0.1����
��5��ʵ������ĺ�����Һ����Ⱦ����������Cr2O72��ת��ΪCr3������ת��ΪCr��OH��3������ȥ��ΪʹCr3��Ũ��С��1��10-5mol/L��Ӧ������Һ��pH=___________������֪��������Cr��OH��3��Ksp��6.4��10-31�� lg2=0.3 lg5=0.7��