��Ŀ����
��֪Ksp(AgCl)=1.8��10��10��Ksp(AgI)=1.0��10��16�����й��ڲ�����֮��ת����˵���д������
| A��AgCl������ˮ������ת��ΪAgI |
| B�������������Ksp���Խ���������Խ����ת��Ϊ�����ܵ������� |
| C��AgI��AgCl��������ˮ������AgCl����ת��ΪAgI |
D�������£�AgCl��Ҫ��NaI��Һ�п�ʼת��ΪAgI����NaI��Ũ�ȱ��벻����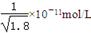 |
A
�������������AgCl������ˮ�����Dz�����ˮ����AgI��������ˮ��ǰ������ת��Ϊ���ߣ���A�������ֲ������Ksp���Խ��Ksp�ϴ���������Խ����ת��Ϊ�����ܵ��������B��ȷ��Ksp(AgCl)>Ksp(AgI)����AgI��AgCl��������ˮ������AgCl����ת��ΪAgI����C��ȷ����NaI��I����Ũ�Ȳ�����xmol/L��AgCl(s) Ag++Cl������AgCl����Һ��c(Ag+)=
Ag++Cl������AgCl����Һ��c(Ag+)=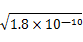 mol/L����c(Ag+)?c(I��)>Ksp(AgI)=1.0��10��16����x>
mol/L����c(Ag+)?c(I��)>Ksp(AgI)=1.0��10��16����x>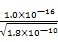 mol/L=
mol/L=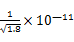 mol/L��AgCl����ת��ΪAgI����D��ȷ��
mol/L��AgCl����ת��ΪAgI����D��ȷ��
���㣺�������ܵ���ʵ��ܽ�ƽ�⣬��Ҫ�漰�ܶȻ���������ת�����ɡ�Ũ�������ܶȻ��Ĺ�ϵ��֪ʶ��

25��ʱ�����и���ֱ��ɵ����0.1 mol��L-1��������Һ��ϣ��йػ�Ϻ����Һ˵������ȷ���ǣ� ��
| A��Ba��OH��2��Һ��Na2SO4��Һ��pH="13" |
| B��������CH3COONa��Һ��c��Na+��+c��H+��= c��CH3COO-��+c��OH-�� |
| C��Na2CO3��Һ��NaHCO3��Һ��2c��Na+�� = 3c��CO32-��+3c��HCO3-��+3c��H2CO3�� |
| D��Na2CO3��Һ��NaOH��Һ�� c��Na+��>c��CO32-��>c��OH-��>c��H+�� |
�����£�����˵����ȷ����
| A����CH3COOH��Һ�м�ˮ��c(OH��)��c(H+)������ |
| B�����ʵ���Ũ�Ⱦ�Ϊ0.1mol?L��1��CH3COOH��Һ��NaOH��Һ�������ϣ�c(Na+)>c(CH3COO��) |
| C��ijŨ�ȵ��Ȼ����Һ�д���c(NH4+)>c(Cl��)>c(H+)> c(OH��) |
| D����pH=4���Ȼ����Һ��c(H+)+ c(NH4+)=" c(Cl��)+" c(OH��) |
25��ʱ����������ȷ����
| A��0.10mol��L��1��������Һ����NaCl����NaOH����CH3COONa����pH��С˳��Ϊ���ۣ��ڣ��� |
| B��0.10mol��L��1NaHCO3��Һ�У�c(CO32��)+c(HCO3��)+c(H2CO3)=0.10mol��L��1 |
| C��pH=2�������pH=12��NaOH��Һ�У�ˮ�����ӻ�Kw��ͬ |
| D���������ͬ��pH=2������ʹ�����ȫ��Ӧ����Ҫ0.010mol��L��1 NaOH�������ͬ |
�����£�ʵ������0.01mol?L��1���ᣬ����������ȷ����
| A��c (CH3COO��) + c (OH��)=" 0.01" mol?L��1 |
| B��������pH = 12�İ�ˮ��Ϻ�������Һ�Լ��� |
| C���������������ƹ����������Һ��c (CH3COO��)��С |
| D��������0.01 mol?L��1NaNO3��Һ��Ϻ��У�c (NO3��) =" c" (CH3COO��)+c (CH3COOH) |
����������pH=3�Ĵ��ᣬ����������ȷ����
| A����ˮϡ�ͺ���ĵ���̶�������Һ�ĵ�������ǿ |
| B��c(H+)=c(CH3COO��)+c(OH��) |
| C���������������ƹ���ָ������£���Һ��c(CH3COO��)?c(H+)/c(CH3COOH)���� |
| D��������pH=11��NaOH��Һ��Ϻ�pH=7 |
�����£�ijŨ�ȵĴ�����Һ��n(CH3COO��)=0.01mol������������ȷ����
| A����ô�����Һ�м���������Ũ�ȵ����ᣬ���ƴ���ĵ��룬��Һ��c(H+)���� |
| B����������Һ���Ϊ1L����c(CH3COOH)=0.01mol/L |
| C����NaOHǡ���к�ʱ����Һ��c(Na+)��c(CH3COO��) |
| D����������Ũ�ȵĴ�������Һ��ϣ���Һ��c(Na+)+ c(H+)=c(CH3COO��)+ c(OH��) |
����˵����ȷ����
A����֪0.1 mol��L-1�Ĵ�����Һ�д��ڵ���ƽ�⣺CH3COOH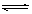 CH3COO����H�����������ռ���Һ��ʹ��Һ�� CH3COO����H�����������ռ���Һ��ʹ��Һ��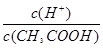 ֵ���� ֵ���� |
B��25��ʱ����ˮ�м�����������CH3COONa��ˮ�ĵ���ƽ�⣺H2O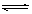 H++OH-�����ƶ���c(H+)���� H++OH-�����ƶ���c(H+)���� |
| C��ȡc(H+)="0.01" mol��L-1������ʹ����100 mL���ֱ�ϡ��2�����ٷֱ����0.03 gп�ۣ�����ͬ�����³�ַ�Ӧ��������п��Ӧ�����ʴ� |
| D������������Һ�еμ�ϡ����õ���pH��5�Ļ����Һ�У�c(Na��)��c(NO3��) |