��Ŀ����
��6�֣���һʵ����ʵ������������ͭ��Һ��Ӧ��������ͭ���ɣ������н϶�����������Ϊȷ������ijɷ֣���������ʵ��̽����
��һ��̽�������������Ԫ�ؽǶȷ��������������SO2��O2��H2�е�һ�ֻ��֡�
������ʵ�飺
��1����ͬѧΪȷ���Ƿ���SO2����������ͨ�����Ը��������Һ�У���Һ��ɫδ�����仯����������� SO2����С����ޡ�����
��2����ͬѧ��Ϊֻ��O2������� �����顣ʵ��֤����ͬѧ�Ĺ۵㲻��ȷ
��3����ͬѧΪ�ж��Ƿ���O2��ͬʱȷ������ɷ֣�ͨ���������ϵ�֪��
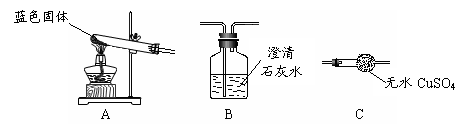
O2+4KI+4HCl ="==" 2I2+4KCl+2H2O��I2Ϊ�ⵥ�ʡ���������������µ�ʵ��װ�ã�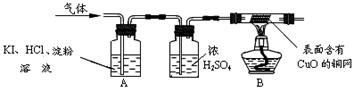
������ʵ����������ۣ�A�й۲쵽 ��֤����O2��
B�й۲쵽 ��֤����������H2��
���ģ�ʵ�鷴˼��
��4�������Ϊ��ͬѧ�ڼ���֮ǰ��Ӧ�������� ��ȷ����ȫ��
��5��������ʵ���֪������ͭ��Һ�� �ԣ���ᡱ�����
��1���ޣ�2�������ǵ�ľ����(��)������������ɫ������ ��4���鴿��5������
���������������1����������������ʹ���������Һ��ɫ�����������Һ��ɫ���䣬˵�������в�����ʹ���������ɫ�Ķ����������壻��2��ʵ����ͨ��ʹ�ô����ǵ�ľ��������������ľ������ȼ����˵�����岻Ϊ������������������û������ʱ����Һ�е�KI�Ͳ��ܷ�����Ӧ���ɵ��ʵ⣬û�е��ʵ����ɵ�����Һ�Ͳ�������ɫ�������ڼ�����������������ͭ��Ӧ����ͭ��ˮ����ˣ����۲쵽��ɫͭ˿��ɹ����ĺ�ɫʱ�����˵������Ϊ��������4�������������ڼ��Ȼ��ȼʱ�����ܻ���ֱ�ը����ˣ���ʹ�������ȿ�ȼ������ǰһ��Ҫ��������Ĵ��ȣ�
��5�����Ļ�Դ�����֮ǰ�������ᷢ���û���Ӧ�ų��������������ݷ�Ӧ�в�������������ʵ�����Ʋ�����ͭ��Һ�����ԣ�
���㣺��������ʵ��֤����������ļ�������ӷ�������Ļ�ѧ���ʣ�������Ӧ��������ը�Ĵ�ʩ

��6�֣����ۡ��ܵ�ͨ����ʹ�����Ĺܵ���ͨ��С��ͨ��ʵ��ԡ��ܵ�ͨ���е����ʳɷֽ�����̽����
������⣺���ܵ�ͨ���к���ʲô���ʣ�
�������ϣ�
��1����ˮ������ٻ�����ҪΪ���ۡ�ʳ�������ë���ȡ�
��2������������ǿ���ȥ�����ۡ���ʴë���ȣ���������ͨ�ܵ���
��3������Ϊ����ɫ���壬�����ᡢ����Һ���ܷ�Ӧ�ų����������ų������ȡ����磬�������ᷴӦ�Ļ�ѧ����ʽ�ǣ� ����������������Һ��Ӧ�Ļ�ѧ����ʽ�ǣ�2Al+2NaOH+ �T2NaAlO2+3H2��
��������֤��С����һ�С��ܵ�ͨ��������һ����ɫ���������һ������ɫ��ĩ��
�����ɫ�������Ϊ�������ơ�ʵ�鷽�����±���ʾ��
| ʵ����� | ʵ������ |
| �ٽ�������ɫ����������ڱ������ϣ�����һ��� | |
| �ڽ�������ɫ�����������ʢ������ˮ���Թ��У������ִ����Թ���� | |
| ��������ڵõ�����Һ�з���һ����ë�ߣ�����һ��ʱ�� | ��ë������ʧ |
���������ɫ��ĩΪ���ۡ�
ʵ�飺ȡ�÷�ĩ�ֱ�������֧�ྻ���Թ��У���һֻ�Թ��еμ�ϡ����,��һֻ�еμ� ��Һ�������������������Թ�����¶��������ߣ���ȼ��Ӧ���������������ʵ���ɫ��
���ۣ�����ɫ��ĩΪ���ۡ�
�����뷴˼����1����ʦ����С�����������غ��������Ƶ����ʷdz����ƣ���ˣ�Ҫȷ�ϲ�����Ƿ���ȷ������Ҫѧϰ��μ�����Ԫ�صĴ��ڣ�
��2��ʹ�ùܵ�ͨʱ���Ƚ���������ɫ��ĩ�����ڱ������Ĺܵ��У�Ȼ���ټ����ɫ�������������һ��ˮ���������Ϲܵ��ڣ��ܵ���Ѹ�ٷ�����Ӧ����д��һ��ʹ�á��ܵ�ͨ��ʱ��ע�����
��
������ճ��ر���Ʒ֮һ������Ҳ�ǻ�����Ⱦ��һ����Ҫ��Դ��������ij��ȤС�����÷Ͼ�п�̸ɵ����Ϊԭ�ϣ����������̽���Ĺ��̡�
��֪ʶ������
��1��п�̵�صĹ������ɣ���ͼ����
��2���ᾧˮ������������ʣ����������¶������£�
�ᾧˮ������ʧȥ���ֻ���ȫ���ᾧˮ������ɫ�ĵ���
���壨CuSO4 ?5H2O������ʱ��ʧȥ�ᾧˮ��Ϊ��ɫ����
ˮ����ͭ��ĩ��CuSO4����
�����Ʊ�𩷯���壨ZnSO4?xH2O��
С��ͬѧ�ι���ij���շϾ�п�̵�صĹ���������չ���������ͼ��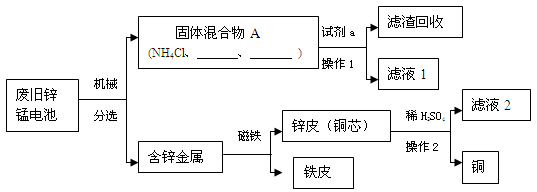
����Һ2����Ũ������ȴ�ᾧ�ɵõ�𩷯���壨ZnSO4?xH2O)������ʱҪ��Ҫ�ò��������Ͻ��裬Ŀ���Ƿ�ֹ ����Һ�ηɽ���
������Ȥ��չ�������ն�������
ʵ�飨���в��ֲ���ɻ��սϴ����Ķ������̹��塣
 ��1��С��ͬѧ��Ϊ�Լ�aΪˮ������Ϊ�ܲ���ʵ��ʵ��Ŀ�ģ��� ��
��1��С��ͬѧ��Ϊ�Լ�aΪˮ������Ϊ�ܲ���ʵ��ʵ��Ŀ�ģ��� ��
��2���ڿ����г�����յ�Ŀ���� ��
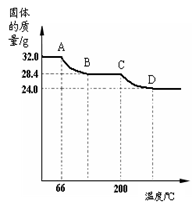
����𩷯�����нᾧˮ�����IJⶨ
С��ͬѧ������𩷯���壨ZnSO4?xH2O������ʵ���ң�����ͼװ�òⶨ�����нᾧˮ�ĺ�����ͼ�а�Ĥ��������ͨ���ֿɷ�ֹ�����ĩ���뵼�ܣ���
�ⶨ��������ȡ28��7g��������Cװ�õ�Ӳ�ʲ������У���������ȫʧȥ�ᾧˮ:
��ZnSO4?xH2O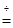 ZnSO4 + xH2O������ȴ�����º����������й���������Ϊ16��1g��
ZnSO4 + xH2O������ȴ�����º����������й���������Ϊ16��1g��
��1��B�е��Լ��ɴ�����������ѡȡ�����ѡ���� ��
| A��Ũ���� | B����������Һ | C������̼������Һ | D������ʯ��ˮ |
��3������ʵ����������𩷯�����нᾧˮ��xֵΪ ��
��9�֣���������ۺ����ۡ�����ۡ��۸����ܴ���ۡ��ָ�û�����Բ��죬ij��ѧ��ȤС��ͬѧ�Զ��߳ɷֵIJ�ͬ������̽����
���������ϡ�
�����������������ӹ����ɣ�����һ������̼��ơ�������ȣ�����������ܽ���ˮ������Ũ���Ტ���Ȼ���ֻ�ɫ���ɫ��
�����ۡ�����ۡ��ɱ��Ǽӹ����ɣ��ӹ������л��õ��������ƣ����ǵ���Ҫ�ɷ���̼��ơ�
����������ۺ����ۡ�����ۡ��У���̼����⣬�����ɷ־����������ᷴӦ���ɶ�����̼��
��������롿��������ۺ����ۡ�����ۡ�����������У�
�����ۡ�����ۡ��к��� ��
�����ۡ�����ۡ��в��������ᡣ
����������ۺ����ۡ�����ۡ���̼��Ƶ�����������ͬ��
��ʵ���������������ۺ����ۡ�����ۡ��ֱ������ֻ�ձ��У�������ˮ�ܽ⣬����һ��ʱ�����ˣ�ȡ������Һ���á�
| ���� | ���� | ���� |
| ȡ����������Һ�ֱ�����֧�Թ��У��μ����� | ��������۵���Һû�б�ɫ�����ۡ�����ۡ�����Һ��� | ����ٳ��� |
| ȡ����������Һ�ֱ�����֧�Թ��У�����һ���� ������������ | ��������۵���Һ�л�ɫ���֣��ֲ���ڣ����ۡ�����ۡ�����Һû���������� | ����ڳ��� |
| | ��������� | ���ۡ�����ۡ� |
| �����ĩ������ | 10.00 g | 10.00 g |
| ����ϡ��������� | 46.00 g | 50.13 g |
| ��Ӧ���ձ������ʵ����� | 52.00 g | 55.77 g |
����д��������̣����������0.1%��
�ó����ۣ����ۡ�����ۡ���̼��Ƶ��������� ��������ۣ�ѡ����ڡ��������ڡ����ڡ�����
����˼�����ۡ���֪ʵ���������ϡ�������������Ϊ14.6% ��С��������ü���ϡ�����������������������������μӷ�Ӧ���Ȼ�����������������û�ѧ����ʽҲ��������ۡ�����ۡ���̼��Ƶ�����������ΪС����˼·�Ƿ���У� ��ѡ����С������С�������������� ��
��5�֣���ѧʵ������һƿ��ɫ����X��Һ�����ǩ�����䣬ij��ȤС�����ͨ��̽��ʵ��ķ�ʽȷ���仯ѧ�ɷݡ�����ȡ��6��X��Һ���ֱ�μ�����6����Һ��ʵ���������±���ʾ�����ƶ�X�����Ǻ����ʣ���д�������Ļ�ѧ����ʽ��
| �ٵμӷ�̪��Һ������������ | �ܵμ�Na2CO3��Һ��������ɫ���� |
| �ڵμ�HClҺҺ������������ | �ݵμ�Na2SO4��Һ��������ɫ���� |
| �۵μ�NaOH��Һ������������ | �μ�AgNO3��Һ��������ɫ���� |
��2��д���ܵĻ�ѧ����ʽ ����3��д���Ļ�ѧ����ʽ ��
��8�֣�ʵ����ϣ���ͬѧ��ʯ����Һ�ⶨϡ�����NaOH��Һ������ԣ���ͬѧ��ʯ����Һ�ⶨʯ��ˮ��Na2CO3��Һ������ԣ�����ͼ��ʾ����ʵ�����з�Һ��������ͬѧ����֧�Թ��е�Һ�嵹���ձ�A�У��õ���ɫ��Һ����ͬѧ����֧�Թ��е�Һ�嵹���ձ�B�У��õ���ɫ��Һ�Ͱ�ɫ���������ս�A�е���Һ��B�е��ϲ���Һ�����ձ�C�С�
��1����ͬѧ��ʵ������ �еμ�ʯ����Һ����Һ��ɫΪ��ɫ��
��2������ͬѧ����֧�Թ��е�Һ���Ϻ��ձ�A�е���Һ����һ���� ��
A��HCl B��NaOH�� C��NaCl
��3����λͬѧ�������ձ�C�еķ�Һ�ɷֽ���̽����
����������衿ͨ����������ʵ�������ձ�C�еķ�Һ��ʯ���ˮ�⣬����ɿ���Ϊ��
���� ���� ��
��������ʵ�顿�����ʵ����֤�ձ�C�еķ�Һ���ܵ���ɣ�
| �������� | ʵ������ | ʵ����� |
| | | |
��4�������λͬѧ���ձ�C�м��� ��ʵ���˴˴�ʵ�����ɫ�ŷţ������ԣ���