��Ŀ����
��������AlN����һ�����������ϣ�ij��������Ʒ�к���̼�����������ʣ���֪��������NaOH��Һ�ܷ�Ӧ���䷴Ӧ����ʽΪ����AlN+NaOH+H2O=NaAlO2+NH3������������Ҳ��������������Һ��Ӧ��ȫ���ܽ⣬�����������壮
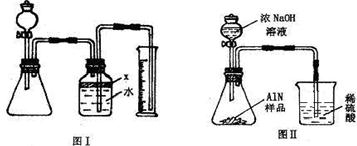
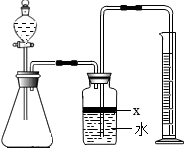
����ͼ���е�һЩװ�����������ʵ�飬���ݷ�Ӧ�������ɰ�����������ⶨ��Ʒ�еĵ�����������������������ʵ��������ȷ�����ʵijɷ֣�ʵ���е���������Բ��ƣ�

��1��ָ��ͼ�������������ƣ�A
��2��ʵ�����Ϊ��a����B�з���������AlN��Ʒ��b��A����ƿ�м��������ŨNaOH��
c������װ�������ԣ�d���ⶨ�ռ���ˮ���������ȷ�IJ���˳��Ϊ
��3���������м��װ�������Եķ�����
��4�����ƿ�е��Լ�X��ѡ��
��5��ʵ����������۲쵽B�л��й��壬����Ʒ�к��е�������
��6����ʵ���в����Ʒ������Ϊ2.56g�����������Ϊ1.12L������£�������Ʒ��AlN����������Ϊ
��7�����˸���ͼ��װ�ý���ͬ��ʵ�飬ͨ���ⶨ�ձ��������������ȷ����Ʒ��AlN����������������Ϊ�Ƿ���У�
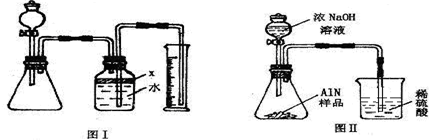
����ͼ���е�һЩװ�����������ʵ�飬���ݷ�Ӧ�������ɰ�����������ⶨ��Ʒ�еĵ�����������������������ʵ��������ȷ�����ʵijɷ֣�ʵ���е���������Բ��ƣ�

��1��ָ��ͼ�������������ƣ�A
��Һ©��
��Һ©��
�� B��ƿ
��ƿ
����2��ʵ�����Ϊ��a����B�з���������AlN��Ʒ��b��A����ƿ�м��������ŨNaOH��
c������װ�������ԣ�d���ⶨ�ռ���ˮ���������ȷ�IJ���˳��Ϊ
c��a��b��d
c��a��b��d
����a��b��c��d��ĸ��ʾ������3���������м��װ�������Եķ�����
�رշ�Һ©������������ƿ�����ƿ���Ҳർ��ˮ������������ʱˮ����������
�رշ�Һ©������������ƿ�����ƿ���Ҳർ��ˮ������������ʱˮ����������
����4�����ƿ�е��Լ�X��ѡ��
ʳ����
ʳ����
���������г������������ƣ������ƿ��Һ��û��װ�����Ϸ����������ռ䣩��ʵ����NH3�����������
����
����ƫ��ƫС�䣩����5��ʵ����������۲쵽B�л��й��壬����Ʒ�к��е�������
̼
̼
����6����ʵ���в����Ʒ������Ϊ2.56g�����������Ϊ1.12L������£�������Ʒ��AlN����������Ϊ
80.1%
80.1%
��AlN����Է�������Ϊ41��ÿ22.4L��������Ϊ17g������7�����˸���ͼ��װ�ý���ͬ��ʵ�飬ͨ���ⶨ�ձ��������������ȷ����Ʒ��AlN����������������Ϊ�Ƿ���У�
������
������
�����롰���С����������С���������������˵ ��ԭ���������ױ����գ�������������
�������ױ����գ�������������
��������������Ŀ��������Ϣ����ȡ����ʱ��Ϊ��ֹװ��©��Ӧ������װ�ú���������װ�������Լ�飬ȷ��װ�ò�©�������ȼӹ�����Һ���ԭ�����ҩƷ��������������ռ���������رշ�Һ©���Ļ�����װ�����γɷ�ջ����������װ�ý��м��ȣ�װ�����������������������������ã��ͻ�۲쵽���ƿ���Ҳർ��ˮ������������ʱˮ���������䣻�����İ�����������ˮ��Ϊ��ֹ��������ˮ��Ҫ��������ˮ���룬���Ӧѡ�����백���������õ�Һ����Ϊ����Һ������ʵ���Ŀ�����ڲⶨ�������������������ռ����������壬��ˣ����ƿ�ڵ�ԭ�����岻�ڲ�����Χ�ڣ�������������������������������Һ��Ӧ���γ�ƫ��������Һ��ˮ�Ͱ�������ַ�Ӧ���й������ʵIJ�������̼����������������Һ��Ӧ����Ҫ���ݷ�Ӧ�Ļ�ѧ����ʽ���ɲ�����������������μӷ�Ӧ��Ʒ�е�������������Ȼ����AlN�����������������������ᷴӦ��������臨�ʹϡ������Һ�������ӣ������ڷ�Ӧ��Ϊ���Ҷ���ʹϡ���ᵹ����������ձ���������ȷ��
����⣺��1����Һ©���������Һ��ҩƷ����ƿ�dz��õķ�Ӧ�������ʴ�Ϊ����Һ©������ƿ
��2����ȡ����ʱ��Ϊ��ֹװ��©��Ӧ������װ�ú���������װ�������Լ�飬ȷ��װ�ò�©�������ȼӹ�����Һ���ԭ�����ҩƷ��������������ռ���������ʴ�Ϊ��c��a��b��d
��3���رշ�Һ©���Ļ�����װ�����γɷ�ջ����������װ�ý��м��ȣ�װ�����������������������������ã��ͻ�۲쵽���ƿ���Ҳർ��ˮ������������ʱˮ���������䣻�ʴ�Ϊ���رշ�Һ©������������ƿ�����ƿ���Ҳർ��ˮ������������ʱˮ����������
��4�������İ�����������ˮ��Ϊ��ֹ��������ˮ��Ҫ��������ˮ���룬���Ӧѡ�����백���������õ�Һ����Ϊ����Һ������ʵ���Ŀ�����ڲⶨ�������������������ռ����������壬��ˣ����ƿ�ڵ�ԭ�����岻�ڲ�����Χ�ڣ��ʴ�Ϊ��ʳ���ͣ�����
��5��������������������������������Һ��Ӧ���γ�ƫ��������Һ��ˮ�Ͱ�������ַ�Ӧ���й������ʵIJ�������̼����������������Һ��Ӧ���ʴ�Ϊ��̼
��6��������Ҫ���ݷ�Ӧ�Ļ�ѧ����ʽ���ɲ�����������������μӷ�Ӧ��Ʒ�е�����������Ϊ2.05g��Ȼ����AlN����������=
��100%��80.1%���ʴ�Ϊ��80.1%
��7�������������ᷴӦ��������臨�ʹϡ������Һ�������ӣ������ڷ�Ӧ��Ϊ���Ҷ���ʹϡ���ᵹ����������ձ���������ȷ���ʴ�Ϊ�������У��������ױ����գ�������������
��2����ȡ����ʱ��Ϊ��ֹװ��©��Ӧ������װ�ú���������װ�������Լ�飬ȷ��װ�ò�©�������ȼӹ�����Һ���ԭ�����ҩƷ��������������ռ���������ʴ�Ϊ��c��a��b��d
��3���رշ�Һ©���Ļ�����װ�����γɷ�ջ����������װ�ý��м��ȣ�װ�����������������������������ã��ͻ�۲쵽���ƿ���Ҳർ��ˮ������������ʱˮ���������䣻�ʴ�Ϊ���رշ�Һ©������������ƿ�����ƿ���Ҳർ��ˮ������������ʱˮ����������
��4�������İ�����������ˮ��Ϊ��ֹ��������ˮ��Ҫ��������ˮ���룬���Ӧѡ�����백���������õ�Һ����Ϊ����Һ������ʵ���Ŀ�����ڲⶨ�������������������ռ����������壬��ˣ����ƿ�ڵ�ԭ�����岻�ڲ�����Χ�ڣ��ʴ�Ϊ��ʳ���ͣ�����
��5��������������������������������Һ��Ӧ���γ�ƫ��������Һ��ˮ�Ͱ�������ַ�Ӧ���й������ʵIJ�������̼����������������Һ��Ӧ���ʴ�Ϊ��̼
��6��������Ҫ���ݷ�Ӧ�Ļ�ѧ����ʽ���ɲ�����������������μӷ�Ӧ��Ʒ�е�����������Ϊ2.05g��Ȼ����AlN����������=
| 2.05g |
| 2.56g |
��7�������������ᷴӦ��������臨�ʹϡ������Һ�������ӣ������ڷ�Ӧ��Ϊ���Ҷ���ʹϡ���ᵹ����������ձ���������ȷ���ʴ�Ϊ�������У��������ױ����գ�������������
����������������ʵ��̽���⣬���û�ѧ�仯ǰ��Ԫ��������������䣬�����ð�����NԪ�ص�����������Ʒ�е����������������������Ŀ�ṩ����Ϣ����Ͽα���ѧ����֪ʶ���������Ҫע��������裮��������Ҫ������ʵ�����У�

��ϰ��ϵ�д�
 �������ͬ������ϵ�д�
�������ͬ������ϵ�д�
�����Ŀ
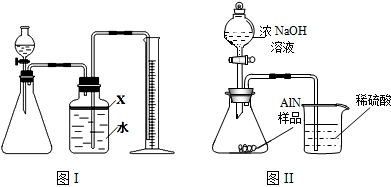
 ��������AlN����һ�����������ϣ��㷺Ӧ���ڼ��ɵ�·��������ij�������к���̼�����������ʣ�ij��ѧ�о���ѧϰС����Ʋ����������ʵ�飮
��������AlN����һ�����������ϣ��㷺Ӧ���ڼ��ɵ�·��������ij�������к���̼�����������ʣ�ij��ѧ�о���ѧϰС����Ʋ����������ʵ�飮