��Ŀ����
����Ŀ����������AlN���������¡�������������Ժõ��������ʣ����㷺Ӧ���ڴ��ģ���ɵ�·�������մɹ�ҵ������ҵ�ϴ��������л������X���ӿ����л�õ������ٽ�����X��N2��̼��һ�������·�Ӧ�Ƶ�AlN��CO����
��1���������մ�����______������ĸ����
A.��������
B.���ǽ�������
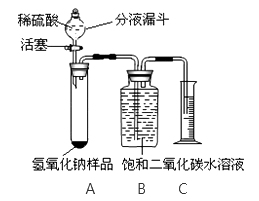
��2��ͨ������£������е������������Ϊ �� ������������������ȥ������̼��ˮ�����Ŀ���Һ����ͨ�������¶Ƚ����������������Һ̬������������������
��3������X�Ļ�ѧʽ�� �� ��AlN�Ļ�ѧ����ʽΪ��
��4�����ڵؿ��к���������Ԫ�صĵ�λ����Ԫ���������������к�����������һ��������������Ԫ������ĺ�����������
���𰸡�
��1��B
��2��78%������
��3��Al2O3��Al2O3+N2+3C ![]() 2AlN+3CO
2AlN+3CO
��4��3����������Ʒ������
����������1���������մ�����B.���ǽ������ϡ���2��ͨ������£������е������������Ϊ78%��������������������ȥ������̼��ˮ�����Ŀ���Һ����ͨ�������¶Ƚ����� ��Һ̬��������������������3������X�Ļ�ѧʽ��Al2O3 �� ��AlN�Ļ�ѧ����ʽΪAl2O3+N2+3C ![]() 2AlN+3CO ��
2AlN+3CO ��
�ʴ�Ϊ����1��B��2��78%��������3��Al2O3��Al2O3+N2+3C![]() 2AlN+3CO��4��3����������Ʒ�����ߣ�1�����ݽ������ϵĶ�����
2AlN+3CO��4��3����������Ʒ�����ߣ�1�����ݽ������ϵĶ�����
��2�����ݿ���������ijɷֽ��
��3�����������غ㶨�ɵ�ʵ���ٸ��ݻ�ѧ��Ӧ����ʽ����д�������
��4�����ݵؿ���ǰ����Ԫ�صĺ�������