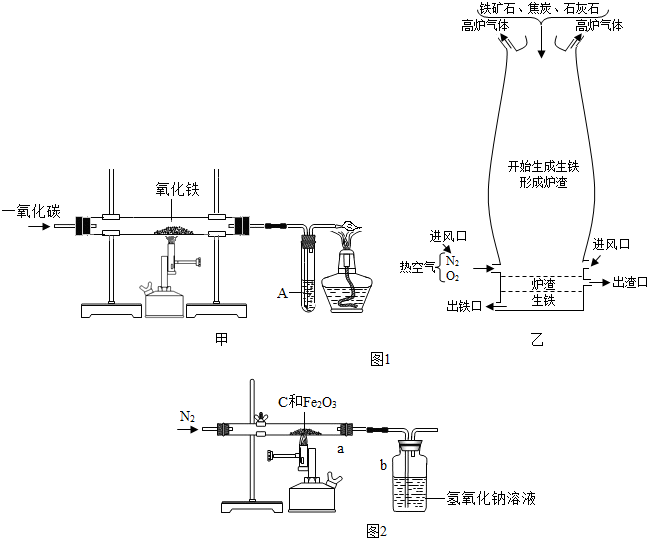
��Ŀ����
Ϊ�˲ⶨij������ʯ��������������������ȡ��ʯ��Ʒ40g������ϡ���ᣬǡ����ȫ��Ӧʱ������ȥϡ����219g�����ˡ�ϴ�ӡ�����������8g����ʯ�е����ʼȲ�����ˮҲ�������ᷴӦ������ش��������⣺��1��д����Ӧ�Ļ�ѧ����ʽ
��2��������ʯ������������������Ϊ
��3���г����μӷ�Ӧ����������x���ı���ʽ
��4����Ӧ��Ĺ�Һ������м���74gˮ����ֽ������ˣ��õ�ֻ��һ�����ʵIJ�������Һ�������ò�������Һ�����ʵ���������Ϊ
��5��������100g����ʵ�����õ�ϡ���ᣬ��Ҫ40%��Ũ��������Ϊ
��������1���������������Ӧ�����Ȼ�����ˮ��
��2����ʯ�е����ʼȲ�����ˮҲ�������ᷴӦ�����ʯ��Ӧ�������ٵ�����Ϊ��ʯ����������������
��3�����������������ᷴӦ�Ļ�ѧ����ʽ���ɷ�Ӧ����������������������μӷ�Ӧ��HCl��������
��4�����������غ㶨��֪����Ӧ����Һ������Ϊ��Ӧ�������������������������֮�ͣ��ɸ��ݵ�������������Ȼ����������������������������
��5���������������غ���м��㣮
��2����ʯ�е����ʼȲ�����ˮҲ�������ᷴӦ�����ʯ��Ӧ�������ٵ�����Ϊ��ʯ����������������
��3�����������������ᷴӦ�Ļ�ѧ����ʽ���ɷ�Ӧ����������������������μӷ�Ӧ��HCl��������
��4�����������غ㶨��֪����Ӧ����Һ������Ϊ��Ӧ�������������������������֮�ͣ��ɸ��ݵ�������������Ȼ����������������������������
��5���������������غ���м��㣮
����⣺��1���������������Ӧ�����Ȼ�����ˮ������ʽΪ��6HCl+Fe2O3=2FeCl3+3H2O
��2��������������Ϊ��ʯ������������������֮�Ϊ40g-8g=32g
������ʯ������������������=
��100%=80%
��3����μӷ�Ӧ����������ʵ�����Ϊx�������Ȼ���������Ϊy
6HCl+Fe2O3=2FeCl3+3H2O
219 160 325
x 32g y
=
=
x=43.8g��y=65g
�������������=
��100%=20%
��4����Ӧ��������Һ������Ϊ219g+32g=251g
����74gˮϡ�ͺ���Һ������Ϊ251g+74g=325g
����������Һ���ʵ���������=
��100%=
��100%=20%
��5������Ҫ40%���������Ϊz
�������������غ�ɵõ�ʽ��100��20%=40%z
��֮�ã�z=50g
�ʴ�Ϊ��
��1��6HCl+Fe2O3=2FeCl3+3H2O
��2��80%����3 ��
=
��4��20%����5��50g
��2��������������Ϊ��ʯ������������������֮�Ϊ40g-8g=32g
������ʯ������������������=
| 32g |
| 40g |
��3����μӷ�Ӧ����������ʵ�����Ϊx�������Ȼ���������Ϊy
6HCl+Fe2O3=2FeCl3+3H2O
219 160 325
x 32g y
| 219 |
| x |
| 160 |
| 32g |
| 325 |
| y |
x=43.8g��y=65g
�������������=
| 43.8g |
| 219g |
��4����Ӧ��������Һ������Ϊ219g+32g=251g
����74gˮϡ�ͺ���Һ������Ϊ251g+74g=325g
����������Һ���ʵ���������=
| y |
| 325 |
| 65g |
| 325g |
��5������Ҫ40%���������Ϊz
�������������غ�ɵõ�ʽ��100��20%=40%z
��֮�ã�z=50g
�ʴ�Ϊ��
��1��6HCl+Fe2O3=2FeCl3+3H2O
��2��80%����3 ��
| 219 |
| x |
| 160 |
| 32g |
��4��20%����5��50g
����������һ��������������Һ���Ʋ�ͬ������������Һ�ļ��㣬�ؼ�������ϡ�������ʲ��䣮

��ϰ��ϵ�д�
�����Ŀ
ʵ����ѧ��ѧ��ȤС��Ϊ�˲ⶨij������ʯ��Fe203������������ȡ20����Ʒ�����ձ��У���ȡϡ�������μ����ձ��У�ÿ�ξ���ַ�Ӧ�����ʲ�����ˮ��Ҳ���μӷ�Ӧ�������ʵ���������£�
| �������� | ��1�� | ��2�� | ��3�� | ��4�� | ��5�� |
| ����ϡ�����������g�� | ����2l | ����21 | ����2l | ����2l | ����21 |
| ʣ������������g�� | ����16 | ����12 | ����8 | ����4 | ����4 |
��2��ǡ����ȫ��Ӧʱ������Һ�����ʵ�����������
ʵ����ѧ��ѧ��ȤС��Ϊ�˲ⶨij������ʯ��Fe203������������ȡ20����Ʒ�� ���ձ��У���ȡϡ�������μ����ձ��У�ÿ�ξ���ַ�Ӧ(���ʲ�����ˮ��Ҳ���μӷ� Ӧ)�����ʵ���������£�
| �� �� | ��1�� | ��2�� | ��3�� | ��4�� | ��5�� |
| ����ϡ���������(g) | 2l | 21 | 2l | 2l | 21 |
| ʣ����������(g) | 16 | 12 | 8 | 4 | 4 |
��1���˳�����ʯ��Fe203������������
��2��ǡ����ȫ��Ӧʱ������Һ�����ʵ�����������