��Ŀ����
�������������������й㷺��Ӧ�ã�
��1��ͼ1��Ʒ�У���Ҫ���ý��������Ե��� ������ĸ����
��2��ů���ܵ�ֹͣ��ů�����ע��ˮ�Է�ֹ�������⣬��ԭ���� ��
��3����������һ����̼��ԭ�ϸ������������Ļ�ѧ����ʽΪ ��
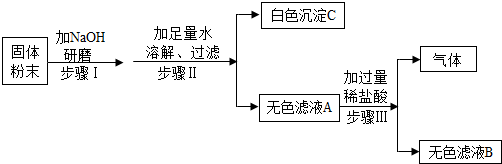
��4����ȤС���ͬѧ��ʵ�������ռ�һͰ����FeSO4��CuSO4�ķ�Һ����������л��ս���ͭ���������ͼ2������
�٢��м���Ľ���X������ ��
�ڢ��м���������H2SO4ʱ���������ݲ�����˵������AΪ ������������������Ӧ�Ļ�ѧ����ʽΪ ���÷�Ӧ���������� ��

��1��ͼ1��Ʒ�У���Ҫ���ý��������Ե���
��2��ů���ܵ�ֹͣ��ů�����ע��ˮ�Է�ֹ�������⣬��ԭ����
��3����������һ����̼��ԭ�ϸ������������Ļ�ѧ����ʽΪ
��4����ȤС���ͬѧ��ʵ�������ռ�һͰ����FeSO4��CuSO4�ķ�Һ����������л��ս���ͭ���������ͼ2������
�٢��м���Ľ���X������
�ڢ��м���������H2SO4ʱ���������ݲ�����˵������AΪ
���㣺�������������ʼ���;,�����Ļ�ѧ����,����ұ��,������ʴ�������������,��Ӧ���͵��ж�,��д��ѧ����ʽ�����ֱ���ʽ�����뷽��ʽ
ר�⣺�������������
��������1�����ݽ����������õĵ����Խ��н��
��2���������������������������ˮͬʱ�Ӵ����н��
��3������һ����̼����������Ӧ�������Ͷ�����̼���н��
��4����������Ŀ���ǻ��ͭ���ʣ�������Ҫ��ͭ����ת��Ϊͭ���ʣ����Լ�����ܹ���ͭ����ת��Ϊͭ���ʵĿ����ǵ��������н��
��2���������������������������ˮͬʱ�Ӵ����н��
��3������һ����̼����������Ӧ�������Ͷ�����̼���н��
��4����������Ŀ���ǻ��ͭ���ʣ�������Ҫ��ͭ����ת��Ϊͭ���ʣ����Լ�����ܹ���ͭ����ת��Ϊͭ���ʵĿ����ǵ��������н��
����⣺��1�������������õĵ����ԣ�����ͭ�����������䵼���ԣ�
��2�����������������������ˮͬʱ�Ӵ�������ů���ܵ�ֹͣ��ů�����ע��ˮ�Է�ֹ�������⣬��ԭ����ʹ�������������������������
��3��һ����̼����������Ӧ�������Ͷ�����̼����ѧ����ʽΪ3CO+Fe2O3
2Fe+3CO��
��4������Ŀ���ǻ��ͭ���ʣ�������Ҫ��ͭ����ת��Ϊͭ���ʣ����Լ�����ܹ���ͭ����ת��Ϊͭ���ʵĿ����ǵ�������
�٢��м���Ľ���X����������
�ڢ��м���������H2SO4ʱ���������ݲ�����˵������A�к�������ͭ��Ϊ�����������ᷴӦ����������������������Ӧ�Ļ�ѧ����ʽΪFe+H2SO4�TH2��+FeSO4���÷�Ӧ�ǵ��ʺͻ����ﷴӦ��������ĵ��ʺͻ�������Ի����������û���Ӧ��
�ʴ�Ϊ����1��B��
��2��ʹ�������������������������
��3��3CO+Fe2O3
2Fe+3CO2��
��4����Fe��
�ڻ���Fe+H2SO4�TH2��+FeSO4���û���Ӧ��
��2�����������������������ˮͬʱ�Ӵ�������ů���ܵ�ֹͣ��ů�����ע��ˮ�Է�ֹ�������⣬��ԭ����ʹ�������������������������
��3��һ����̼����������Ӧ�������Ͷ�����̼����ѧ����ʽΪ3CO+Fe2O3
| ||
��4������Ŀ���ǻ��ͭ���ʣ�������Ҫ��ͭ����ת��Ϊͭ���ʣ����Լ�����ܹ���ͭ����ת��Ϊͭ���ʵĿ����ǵ�������
�٢��м���Ľ���X����������
�ڢ��м���������H2SO4ʱ���������ݲ�����˵������A�к�������ͭ��Ϊ�����������ᷴӦ����������������������Ӧ�Ļ�ѧ����ʽΪFe+H2SO4�TH2��+FeSO4���÷�Ӧ�ǵ��ʺͻ����ﷴӦ��������ĵ��ʺͻ�������Ի����������û���Ӧ��
�ʴ�Ϊ����1��B��
��2��ʹ�������������������������
��3��3CO+Fe2O3
| ||
��4����Fe��
�ڻ���Fe+H2SO4�TH2��+FeSO4���û���Ӧ��
�����������Ĺؼ���Ҫ����˽���������ʣ�ֻ���������ܶ�����������ȷ���жϣ�

��ϰ��ϵ�д�
 ��1����Ԫ�¿�������ĩϵ�д�
��1����Ԫ�¿�������ĩϵ�д�
�����Ŀ
��һ���ܱյ������мס��ҡ��������������ʣ�ͨ�������ȼ��ַ�����Ӧ�����ⶨ�������ڷ�Ӧǰ������ʵ�������ͼ��������Է�������Ϊn��������Է�������Ϊ2n��������˵����ȷ���ǣ�������
| ���� | �� | �� | �� | �� |
| ��Ӧǰ����g | 4 | 10 | 1 | 25 |
| ��Ӧ������g | δ�� | 21 | 10 | 9 |
| A���÷�Ӧһ�����û���Ӧ |
| B���÷�Ӧ�����ɵ��������������Ϊ11��9 |
| C����Ӧ�������Ϊ4g |
| D���÷�Ӧ�м��붡�Ļ�ѧ������֮��Ϊ1��2 |
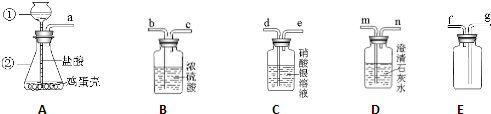
 2013�������г��б�ҵ�ͻ�ѧʵ��������ܿ����У�С���鵽�������ǡ�ˮ�ľ�������������С��һ�����飬���ش��������⣮
2013�������г��б�ҵ�ͻ�ѧʵ��������ܿ����У�С���鵽�������ǡ�ˮ�ľ�������������С��һ�����飬���ش��������⣮