��Ŀ����
����Ŀ���ں�ۡ��ۺͷ���֮�佨����ϵ�ǻ�ѧѧ��ѧϰ���ص㡣
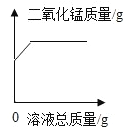
��1���ס��ҡ���������ʾ�������ʣ����ǵ���ʾ��ͼ�����ʾ��
���� | �� | �� | �� | �� |
|
��ʾ��ͼ |
|
|
|
|
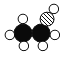
��һ�������ӹ���___________��ԭ�ӡ�
�ڼס��ҡ������������������������������__________��____________��д��ѧʽ����
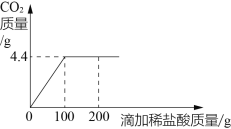
��2����ҵ��ʹ��һ�ֹ������´������������ķ�Ӧ������ͼ��ʾ��
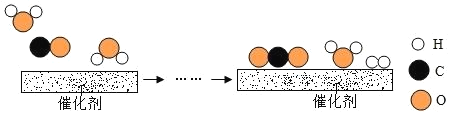
�ٸ÷�Ӧ�Ļ�ѧ����ʽΪ_______________��
�ڲμӷ�Ӧ��![]() ��
��![]() ���Ӹ���֮��Ϊ______________��
���Ӹ���֮��Ϊ______________��
���𰸡�9 ![]()
![]()
 1:1
1:1
��������
��ͼʾ��֪���ױ�ʾ��������һ����̼���ұ�ʾ������������������ʾ���������Ҵ�������ʾ��������ˮ��һ����̼��ˮ�ڵ��´��������ɶ�����̼��������
��1������ͼʾ��֪�������ʵĻ�ѧʽΪ![]() ����һ����������2��̼ԭ�ӡ�6����ԭ�Ӻ�1����ԭ�ӹ��ɣ�����
����һ����������2��̼ԭ�ӡ�6����ԭ�Ӻ�1����ԭ�ӹ��ɣ�����![]() ��ԭ�ӡ�
��ԭ�ӡ�
����ͼʾ��֪���ױ�ʾ��������һ����̼���ұ�ʾ������������������ʾ���������Ҵ�������ʾ��������ˮ���ʼס��ҡ������������������������������һ����̼��ˮ����ѧʽΪ![]() ��
��![]() ��
��
��2�����ɹ�ҵ��ʹ��һ�ֹ������´������������ķ�Ӧ����ͼ��֪����Ӧ��һ����̼��ˮ�ڵ��´��������ɶ�����̼���������ʸ÷�Ӧ�Ļ�ѧ����ʽΪ ��
��
�ڲμӷ�Ӧ��![]() ��
��![]() ���Ӹ���֮��Ϊ1:1��
���Ӹ���֮��Ϊ1:1��

����Ŀ�������ֶ���������̼��ɵĺϽ�������̼�ĺ���Ϊ2%��4.3%������̼�ĺ���Ϊ 0.03%��2%��Ϊ�ⶨij�����Ͻ���̼�ĺ�����С��ͬѧȡһ�������ĺϽ���Ʒʢ�����ձ��У�����������μ���ϡ������ǡ����ȫ��Ӧ��ʵ���������±���ʾ��
�ձ����� | ��Ʒ������ | ϡ��������� | �ձ�+ʣ����������� |
50g | 5.8g | 94.6g | 150.2g |
ע��̼�Ȳ�����ˮҲ����ϡ���ᷢ����Ӧ��
����������Ϣ������м��㣺
��1��ǡ����ȫ��Ӧʱ����������������Ϊ_____ g��
��2����Ʒ��̼����������_____��������������0.1%����
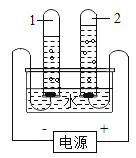
����Ŀ��Ϊ̽������Ļ�ѧ���ʣ�ij��ѧС����������ʵ�飺
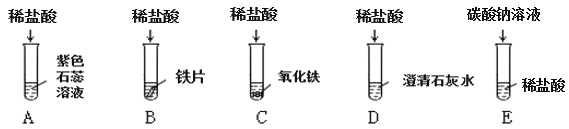
��1��������Ӧ�������������Ϊ_______________������ĸ��ţ���ͬ���������ݲ�������____________��B �Թ�����������Ӧ�Ļ�ѧ����ʽΪ____________��
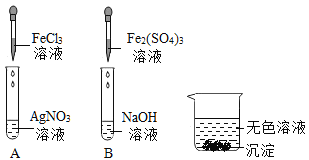
��2������Ӧ�� D �� E �Թ��еķ�Һ����һ�ྻ���ձ��У��۲쵽�ձ����������ݲ��������а�ɫ�������֡����ձ��еĻ������ˣ��õ���ɫ��������ɫ��Һ��ͬѧ�Ƕ���Һ�����ʵijɷֽ��� ̽����
�������ϵ�֪��CaCl2+Na2CO3=2NaCl+CaCO3����Na2CO3+2HCl=2NaCl+H2O+CO2![]() ��
��
��������⣩��Һ�����ʵijɷ���ʲô��
����������룩����һ��NaCl���������NaCl �� CaCl2 ����������NaCl��Ca(OH)2 �� HCl�������ģ�____________��
�������뽻���������ۣ�ͬѧ��һ����Ϊ����____________�Ǵ���ġ�
��ʵ������ۣ�
ʵ�鲽�� | ʵ������ | ʵ����� |
ȡ������Һ���Թ��У��μ�����̼������Һ | ������ | ������ ���� |
ȡ������Һ���Թ��У��μ�����_____________ | ____________ |
����չ��Ǩ�ƣ�ϡ�����ϡ���������ƵĻ�ѧ���ʣ�����Ϊ���ǵ���Һ�ж�����____________��
����Ŀ����ȤС���ͬѧͨ��ʵ��̽����ˮ���ײ�ˮ������Ҫ�ɷ֡�
��������⣩Ӳˮ��к�İ�ɫ���壨ˮ�����к���ʲô���ʣ�
���������ϣ�
��.Ӳˮ�к���һ������ Ca(HCO3)2 �� Mg(HCO3)2������ʱ���������ܽ�ȸ�С�����ʡ� �й����ʵ��ܽ��ԣ�
�� �� | Ca(HCO3)2 | Mg(HCO3)2 | Ca(OH)2 | Mg(OH)2 | CaCO3 | MgCO3 |
�ܽ��� | ���� | ���� | �� | ���� | ���� | �� |
��. Mg(OH)2 �� MgCO3 ���ȷֽ���ֱ����ɶ�Ӧ�����������
�����룩ˮ������Ҫ�ɷ�һ�����в��ܵ�Mg(OH)2 ��________�����ܺ����ܵ�Ca(OH)2��______��
��ʵ�飩
��1������̽����ɫ����ɷ�
����ͬѧȡ��ɫ������������������ˮ����ܽ⣬���ϲ���Һ�еμ�________��Һ��ѡ ����ţ���û�а�ɫ�������ɣ�˵��ˮ������ Ca(OH)2��
A ̼���� B �Ȼ��� C ̼���
����ȡ��ɫ�������ϡ���ᣬ�۲쵽�����ݲ�������ͬѧ��Ϊ������ MgCO3������ͬѧ�������Ľ��ۣ�������__________��
����֪Ca(HCO3)2 ���ȷֽ���̼���������ȷֽ����ƣ���д��Ca(HCO3)2 ���ȷֽ�Ļ�ѧ����ʽ___________________��
��2��Ϊ��һ��ȷ����ɫ����ijɷ֣���ͬѧ��ȡ 20g ��ɫ���壬�������ʵ�飺

��ʯ�ҵ���Ҫ�ɷ��ǹ��� CaO �� NaOH �Ļ���
����Ӧǰ����ͨһ��ʱ��Ŀ�����Ŀ����______________��
��ʵ���� E װ�õ�������____________________��
����ַ�Ӧ��� C ���� 1.8g��D ������ 6.6g��20g ��ɫ������ Mg(OH)2 ��������___________________g��
�����ۣ��ð�ɫ������������þ��̼��Ƶ�������Ϊ_________��